
Rational design of a conformation-specific antibody for the quantification of Aβ oligomers. - Abstract - Europe PMC

An anticancer drug suppresses the primary nucleation reaction that initiates the production of the toxic Aβ42 aggregates linked with Alzheimer's disease | Science Advances

Moxifloxacin Disrupts and Attenuates Aβ42 Fibril and Oligomer Formation: Plausibly Repositioning an Antibiotic as Therapeutic against Alzheimer's Disease | ACS Chemical Neuroscience

PDF) Small molecule-mediated co-assembly of amyloid-β oligomers reduces neurotoxicity through promoting non-fibrillar aggregation
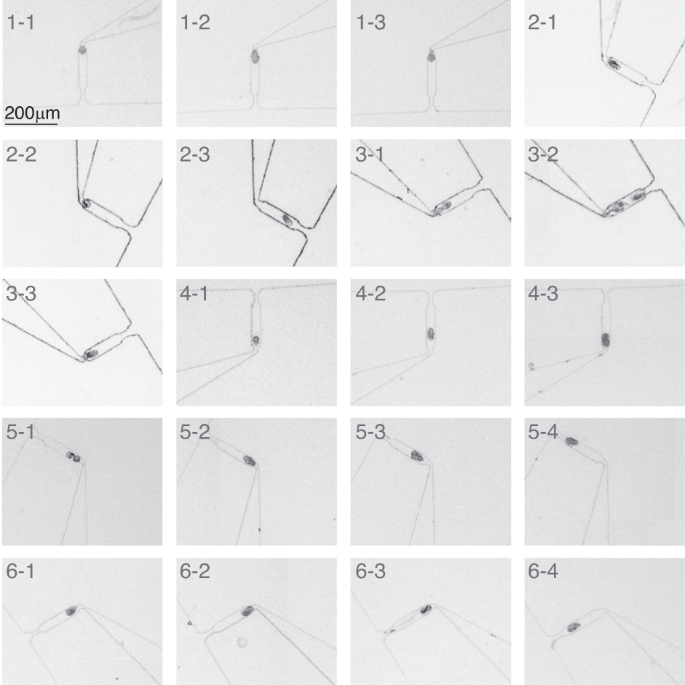
A spiral microfluidic device for rapid sorting, trapping, and long-term live imaging of Caenorhabditis elegans embryos | Microsystems & Nanoengineering

An anticancer drug suppresses the primary nucleation reaction that initiates the production of the toxic Aβ42 aggregates linked with Alzheimer's disease | Science Advances

Perphenazine–Macrocycle Conjugates Rapidly Sequester the Aβ42 Monomer and Prevent Formation of Toxic Oligomers and Amyloid | ACS Chemical Neuroscience

An anticancer drug suppresses the primary nucleation reaction that initiates the production of the toxic Aβ42 aggregates linked with Alzheimer's disease | Science Advances

Rational design of a conformation-specific antibody for the quantification of Aβ oligomers. - Abstract - Europe PMC
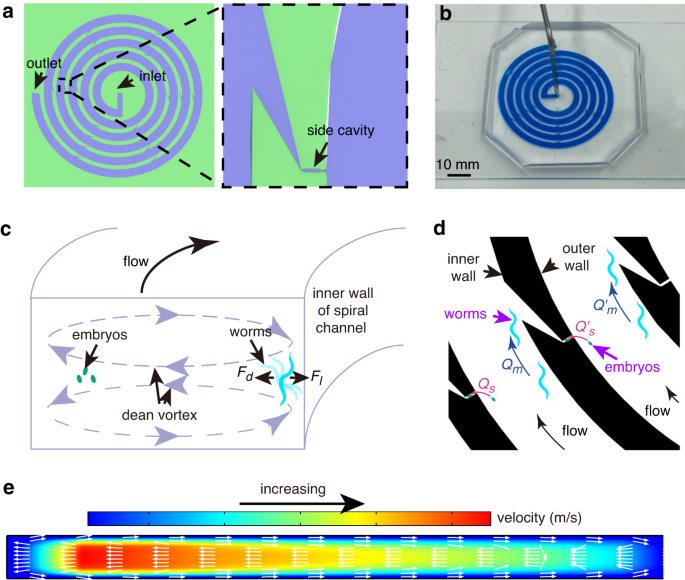
A spiral microfluidic device for rapid sorting, trapping, and long-term live imaging of Caenorhabditis elegans embryos | Microsystems & Nanoengineering
![Hexahydropyrrolo[2,3-b]indole Compounds as Potential Therapeutics for Alzheimer's Disease | ACS Chemical Neuroscience Hexahydropyrrolo[2,3-b]indole Compounds as Potential Therapeutics for Alzheimer's Disease | ACS Chemical Neuroscience](https://pubs.acs.org/cms/10.1021/acschemneuro.9b00297/asset/images/medium/cn9b00297_0007.gif)
Hexahydropyrrolo[2,3-b]indole Compounds as Potential Therapeutics for Alzheimer's Disease | ACS Chemical Neuroscience

Pharmaceuticals | Free Full-Text | Identification of a Thyroid Hormone Derivative as a Pleiotropic Agent for the Treatment of Alzheimer’s Disease

Small-molecule sequestration of amyloid-β as a drug discovery strategy for Alzheimer's disease | Science Advances

Selective targeting of primary and secondary nucleation pathways in Aβ42 aggregation using a rational antibody scanning method | Science Advances
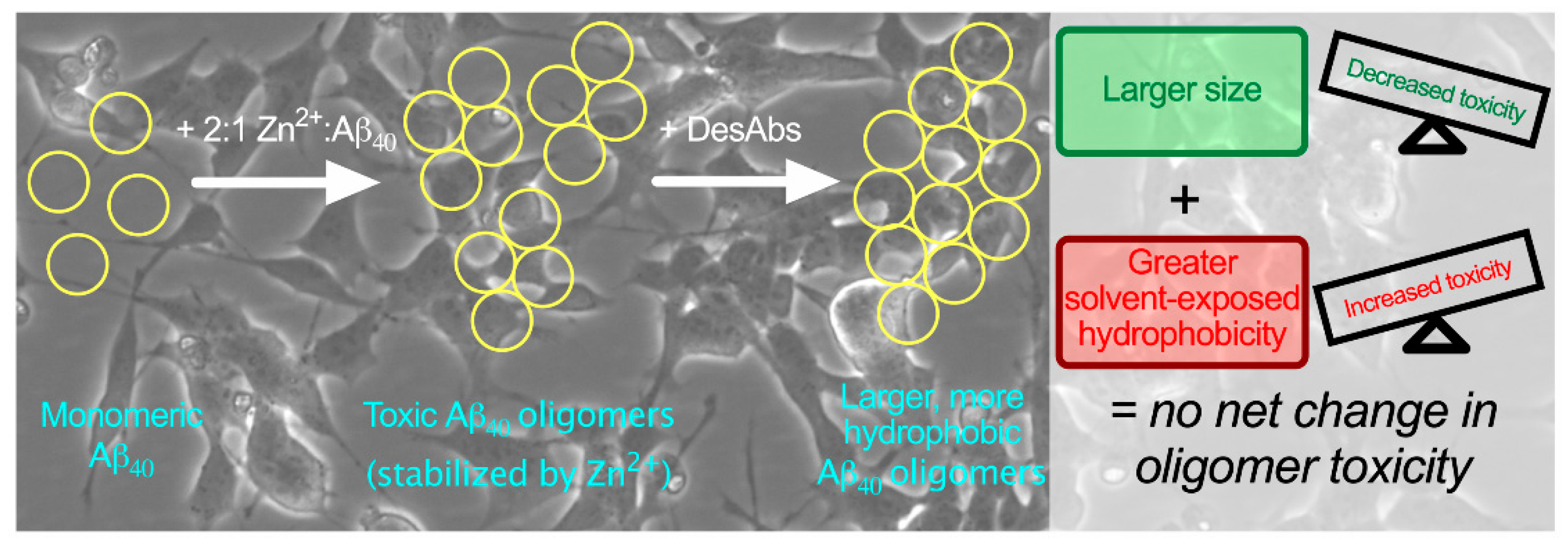
IJMS | Free Full-Text | Rationally Designed Antibodies as Research Tools to Study the Structure–Toxicity Relationship of Amyloid-β Oligomers

PDF) Exogenous misfolded protein oligomers can cross the intestinal barrier and cause a disease phenotype in C. elegans

PDF) A rationally designed bicyclic peptide remodels Aβ42 aggregation in vitro and reduces its toxicity in a worm model of Alzheimer's disease

Perphenazine–Macrocycle Conjugates Rapidly Sequester the Aβ42 Monomer and Prevent Formation of Toxic Oligomers and Amyloid | ACS Chemical Neuroscience

An anticancer drug suppresses the primary nucleation reaction that initiates the production of the toxic Aβ42 aggregates linked with Alzheimer's disease | Science Advances




