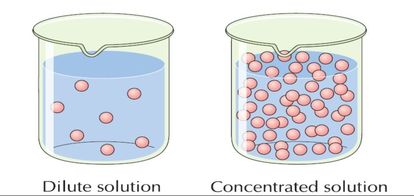
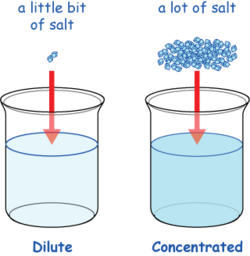
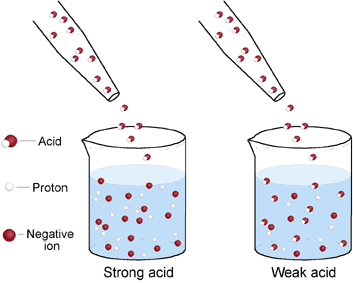
Science - Upper Primary - YDP - Simulation - Neutralisation of alkalis by concentrated and dilute acids

Concentration, normality and amounts of concentrated acids and bases to... | Download Scientific Diagram

A bottle of concentrated sulphuric or sulfuric acid as used in a UK secondary school, London, UK Stock Photo - Alamy
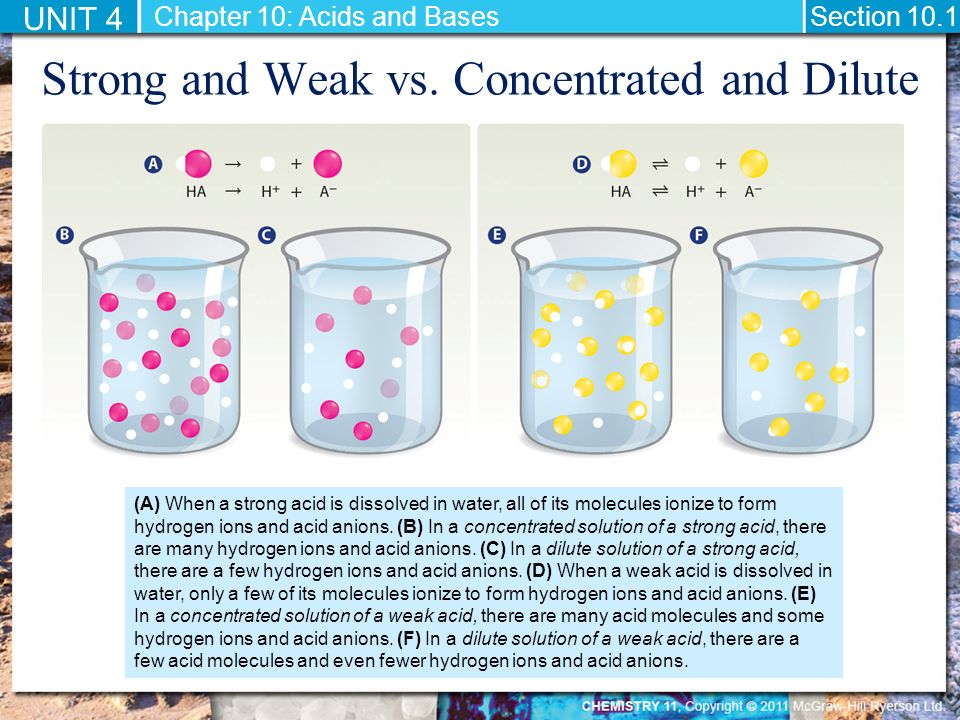
UNIT 4: Solutions: Dilutions & Titrations. Strong Acids An acid that ionizes completely in water is called a strong acid. Hydrochloric acid, HCl(aq), - ppt download