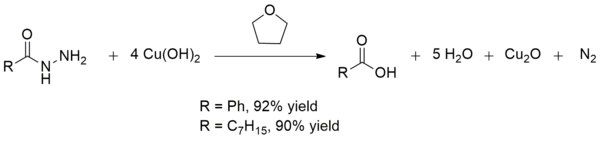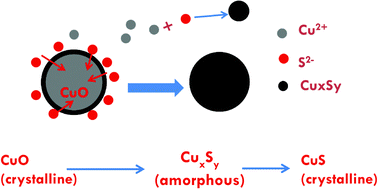
Sulfidation of copper oxide nanoparticles and properties of resulting copper sulfide - Environmental Science: Nano (RSC Publishing)

Copper I and copper II oxides. These two compounds were easily prepared in my lab. Copper II oxide is very easy to make. Basic copper carbonate or Copper hydroxide will easily decompose

Molecules | Free Full-Text | Formation of Copper Oxide Nanotextures on Porous Calcium Carbonate Templates for Water Treatment

Investigating the Origin of Enhanced C2+ Selectivity in Oxide-/Hydroxide-Derived Copper Electrodes during CO2 Electroreduction | Journal of the American Chemical Society
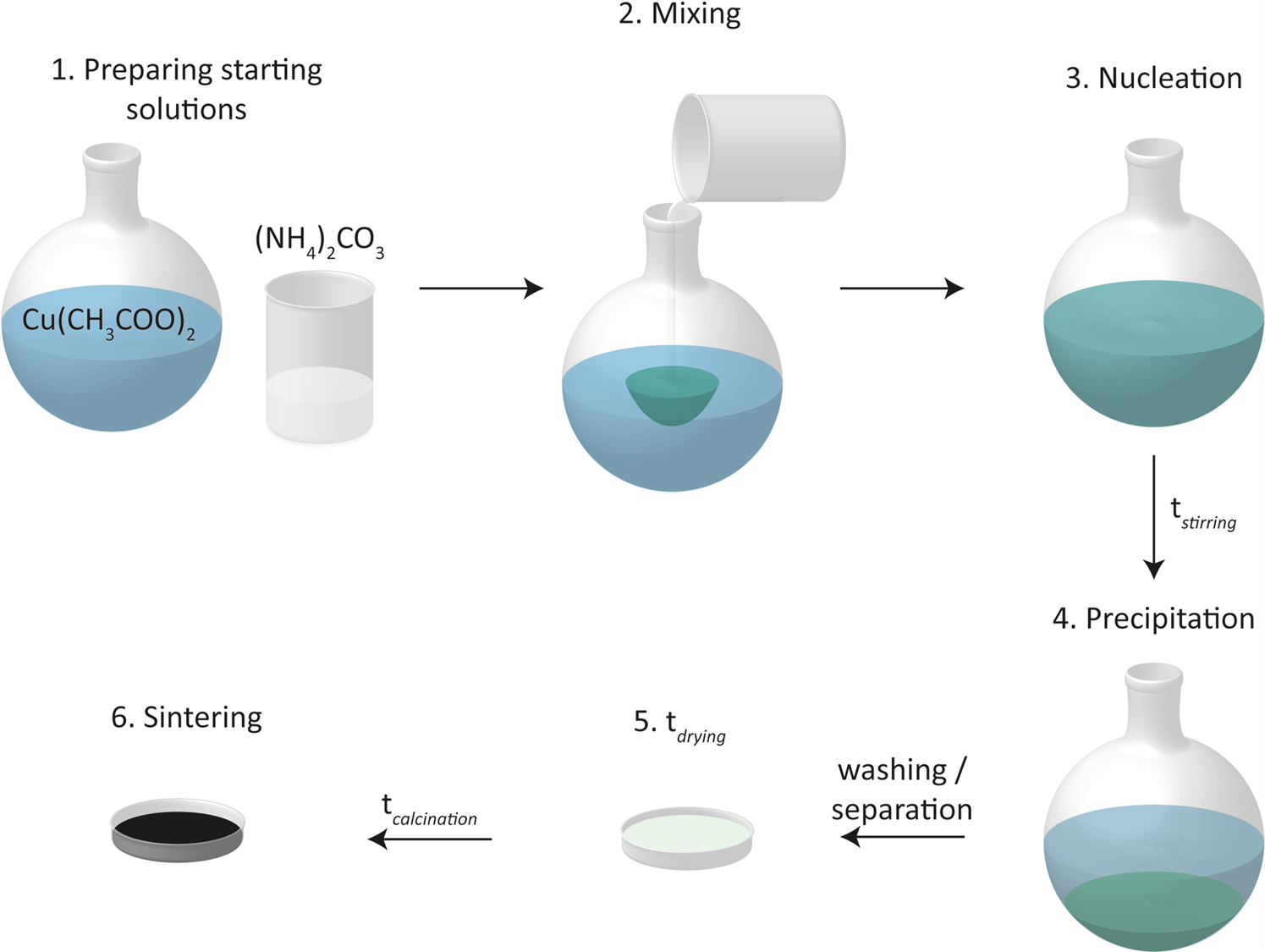
Cost-effective sol-gel synthesis of porous CuO nanoparticle aggregates with tunable specific surface area | Scientific Reports

Bases. Jars containing calcium carbonate (Ca2CO3), copper oxide (CuO) and sodium hydroxide (NaOH). These compounds are classified as bases, because th Stock Photo - Alamy

SOLVED: Post-lh Questionsi Copper (II) hydroxide is converted into copper (II) oxide by heating the test - tube containing Cu (OH) 2 in hot water bath: Is it necessary t0 use distilled

Room-temperature vertically-aligned copper oxide nanoblades synthesized by electrochemical restructuring of copper hydroxide nanorods: An electrode for high energy density hybrid device - ScienceDirect

Kinetics and Mechanisms of the Thermal Decomposition of Copper(II) Hydroxide: A Consecutive Process Comprising Induction Period, Surface Reaction, and Phase Boundary-Controlled Reaction | The Journal of Physical Chemistry C

China Copper Oxide Pricelist – CAS 62-56-6 Thiourea Crystal White Powder on Sale – Fitech Manufacture and Factory | Fitech

Copper oxide formula and copper hydroxide formula and define both of these - Chemistry - Materials Metals and Non-metals - 15135775 | Meritnation.com
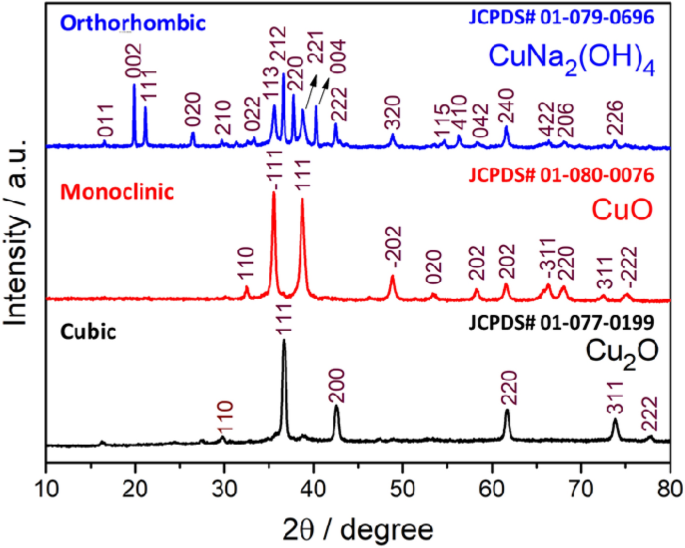
Structural and morphological tuning of Cu-based metal oxide nanoparticles by a facile chemical method and highly electrochemical sensing of sulphite | Scientific Reports