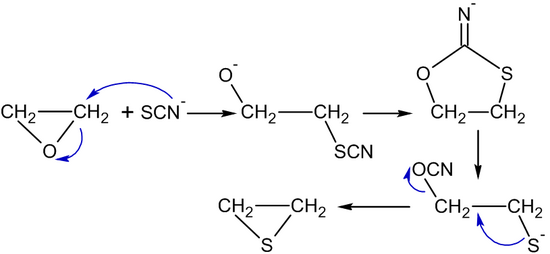
Synthesize the following compound from ethylene oxide and any other reagents of your choice. | Homework.Study.com
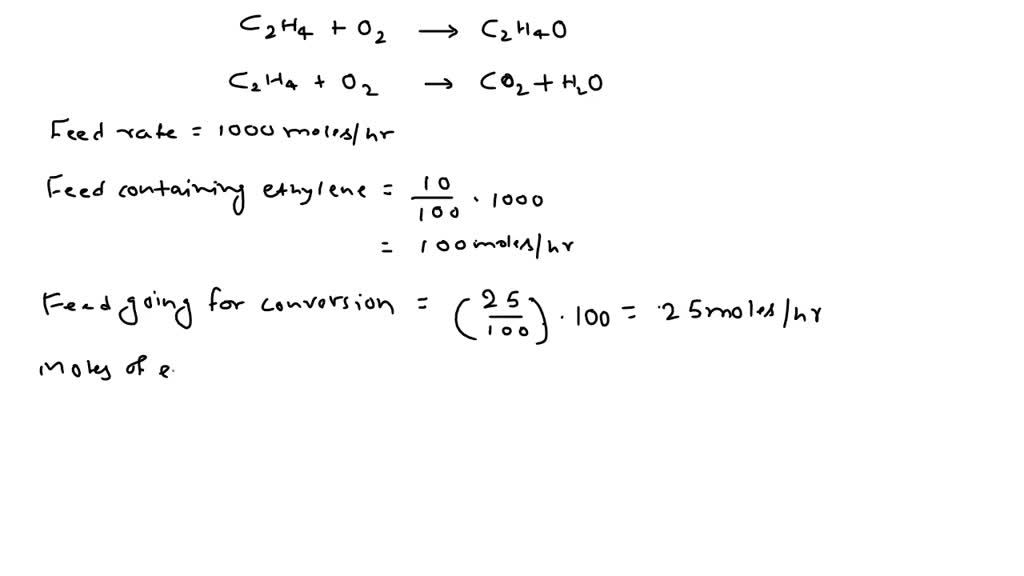
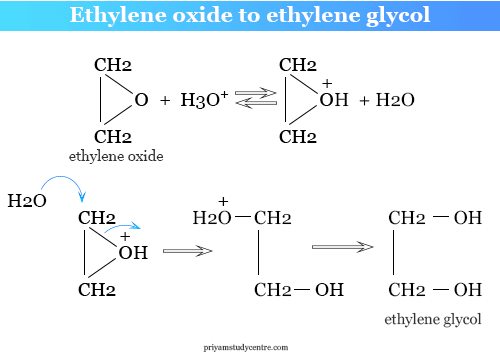
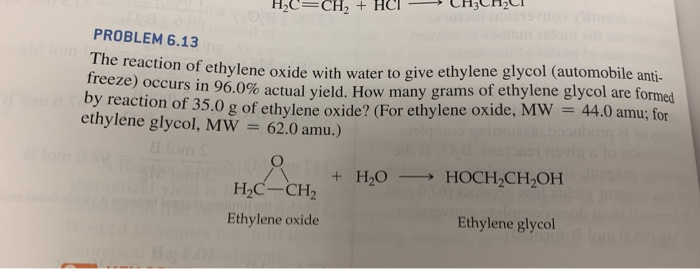
SOLVED: Ethylene glycol (HOCH, CH, OH), used as an antifreeze, is produced by reacting ethylene oxide with water. A side reaction produces an unwanted dimer, DEG: C2H40 + H2O → HOCH2CH2OH HOCH2CH2OH +

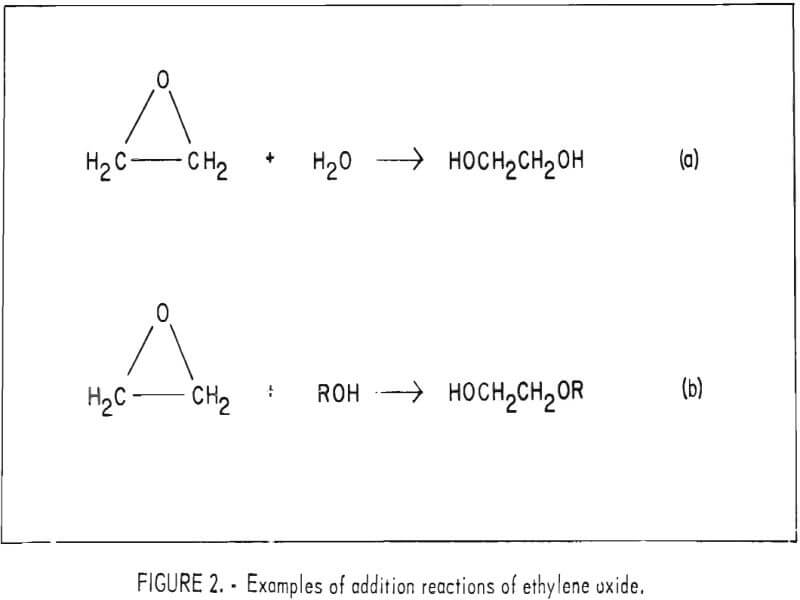
Polymerization of Ethylene Oxide, Propylene Oxide, and Other Alkylene Oxides: Synthesis, Novel Polymer Architectures, and Bioconjugation | Chemical Reviews

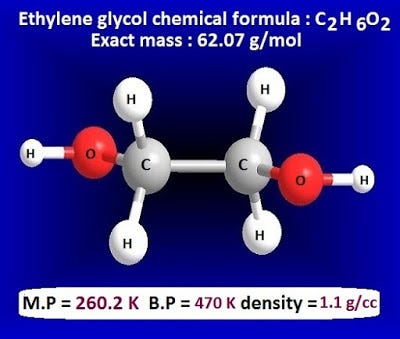
Enthalpies of reaction calculated from bond energies and from enthalpies of formation are often, but not always, close to each other. Ethylene glycol is produced by the catalytic oxidation of the ethy





.jpg)
.jpg)

.jpg)


.jpg)