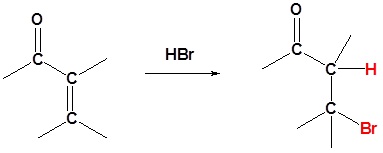
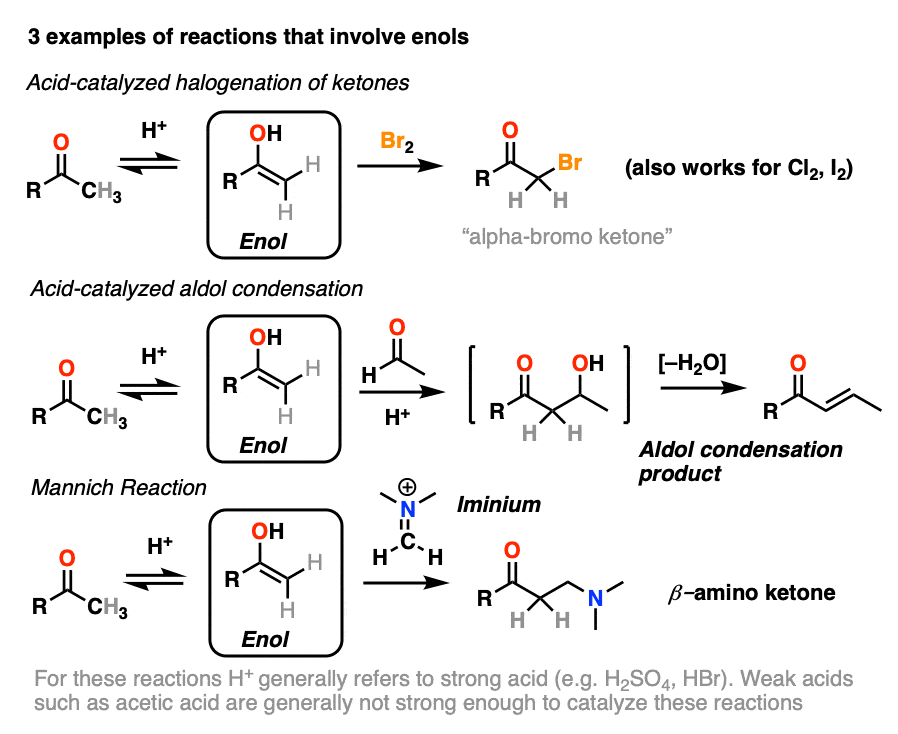
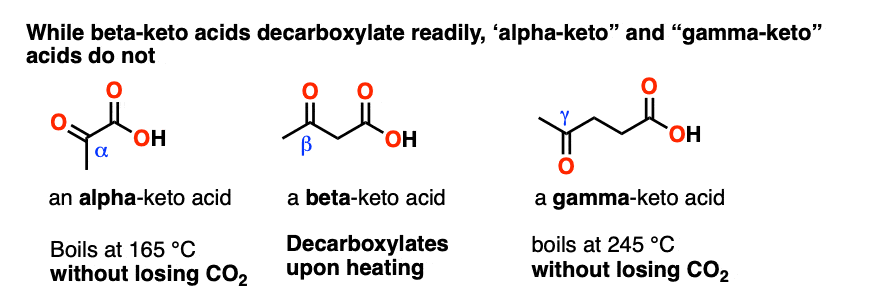A Convenient Halogenation of α,β-Unsaturated Carbonyl Compounds with OXONE® and Hydrohalic Acid (HBr, HCl)
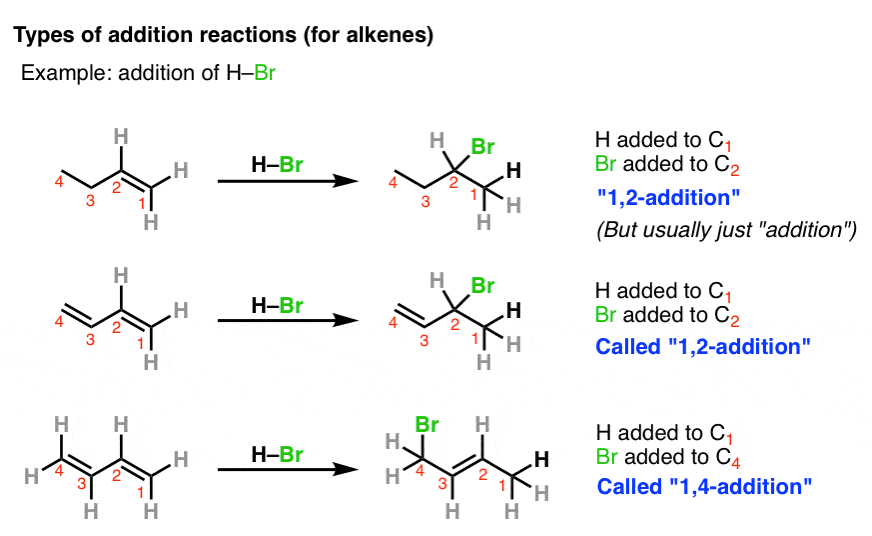
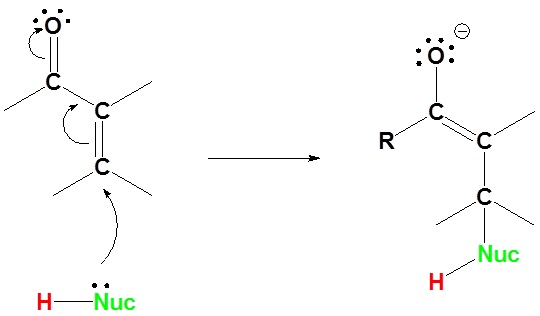
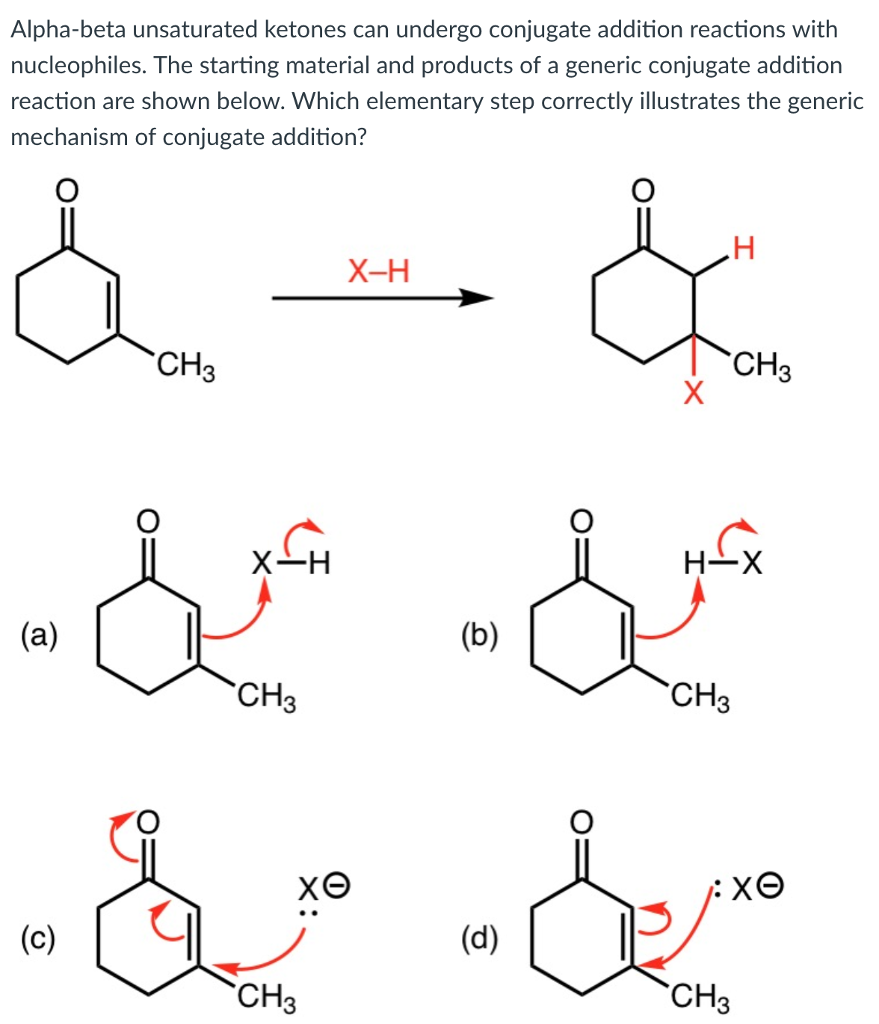
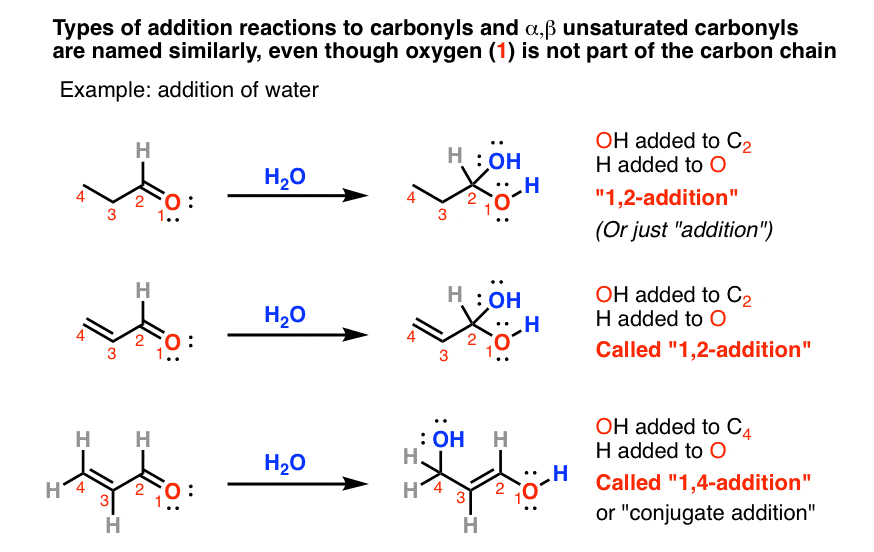
19.13: Conjugate Nucleophilic Addition to alpha, beta-unsaturated Aldehydes and Ketones - Chemistry LibreTexts

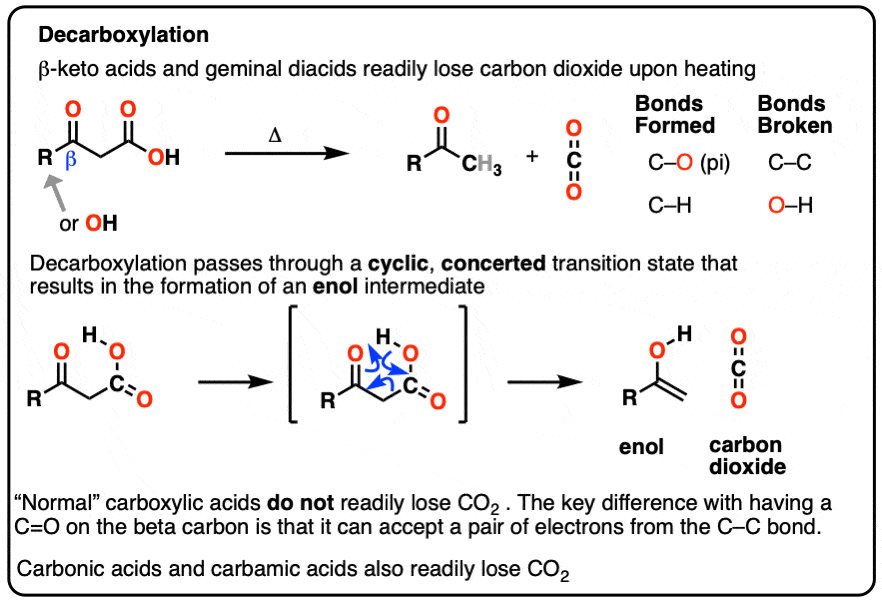
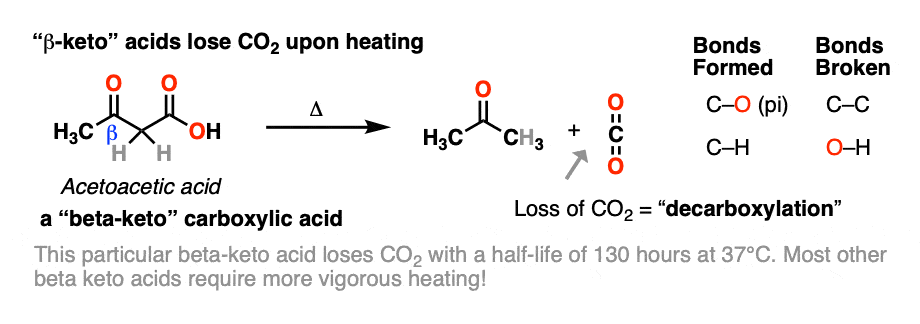
Alkyl Substituted Beta-Keto Acids: Molecular Structure and Decarboxylation Kinetics in Aqueous Solution and on the Surface of Metal Oxides | The Journal of Physical Chemistry C

Organocatalyzed Sulfa-Michael Addition of Thiophenols on Trisubstituted α-Fluoroacrylates, a Straightforward Access to Chiral Fluorinated Compounds | The Journal of Organic Chemistry
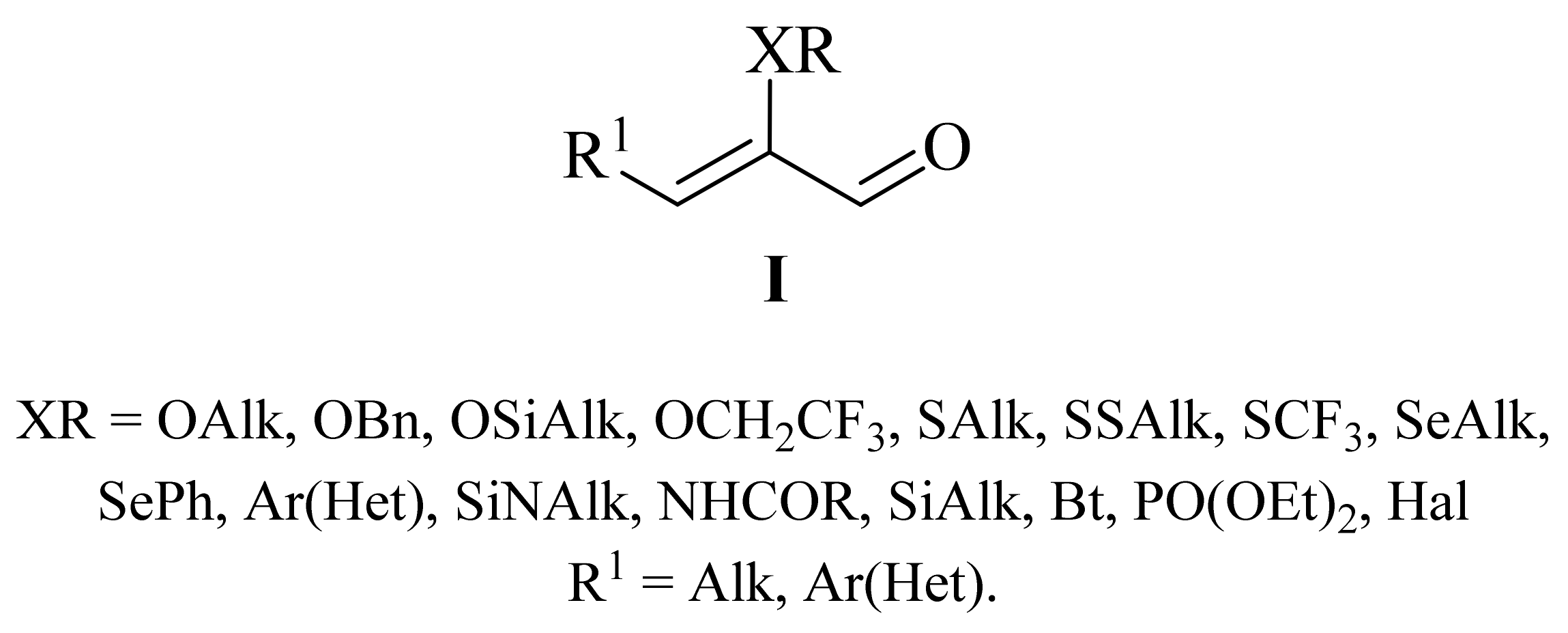
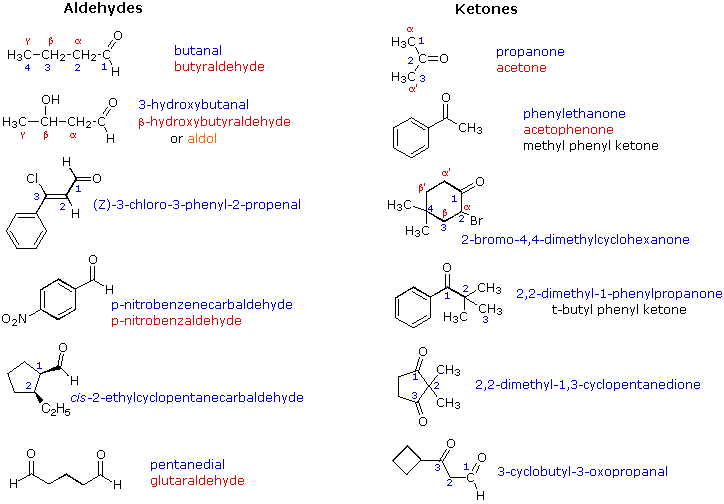
Molecules | Free Full-Text | α-Functionally Substituted α,β-Unsaturated Aldehydes as Fine Chemicals Reagents: Synthesis and Application

Cyclopropylcarbinyl-Type Ring Openings. Reconciling the Chemistry of Neutral Radicals and Radical Anions | Journal of the American Chemical Society
![Multicomponent Domino Reactions for the Synthesis of Variable Hydrogenated Imidazo[1,2‐a]pyridines - Kushch - 2022 - Asian Journal of Organic Chemistry - Wiley Online Library Multicomponent Domino Reactions for the Synthesis of Variable Hydrogenated Imidazo[1,2‐a]pyridines - Kushch - 2022 - Asian Journal of Organic Chemistry - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/b4b74dcb-2348-44c6-b619-11ec445e35da/ajoc202100709-toc-0001-m.jpg)
Multicomponent Domino Reactions for the Synthesis of Variable Hydrogenated Imidazo[1,2‐a]pyridines - Kushch - 2022 - Asian Journal of Organic Chemistry - Wiley Online Library

Synthesis of γ-Oxo-α-amino Acids via Radical Acylation with Carboxylic Acids | The Journal of Organic Chemistry

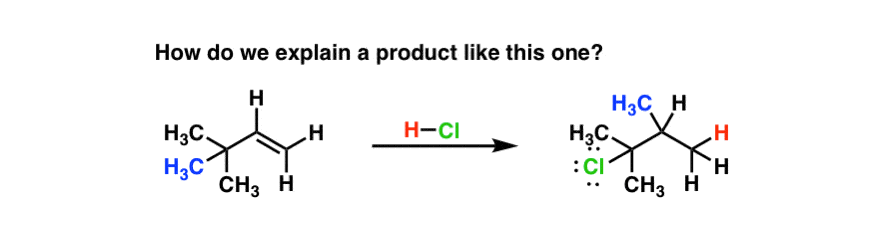
Palladium(II)-Catalyzed Directed anti-Hydrochlorination of Unactivated Alkynes with HCl | Journal of the American Chemical Society
![4‐(Di‐/Trifluoromethyl)‐2‐heterabicyclo[2.1.1]hexanes: Advanced Fluorinated Phenyl Isosteres and Proline analogues - Homon - 2021 - European Journal of Organic Chemistry - Wiley Online Library 4‐(Di‐/Trifluoromethyl)‐2‐heterabicyclo[2.1.1]hexanes: Advanced Fluorinated Phenyl Isosteres and Proline analogues - Homon - 2021 - European Journal of Organic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/854dc161-85b2-4d4a-973d-05bdea214d58/ejoc202100414-toc-0001-m.jpg)
4‐(Di‐/Trifluoromethyl)‐2‐heterabicyclo[2.1.1]hexanes: Advanced Fluorinated Phenyl Isosteres and Proline analogues - Homon - 2021 - European Journal of Organic Chemistry - Wiley Online Library