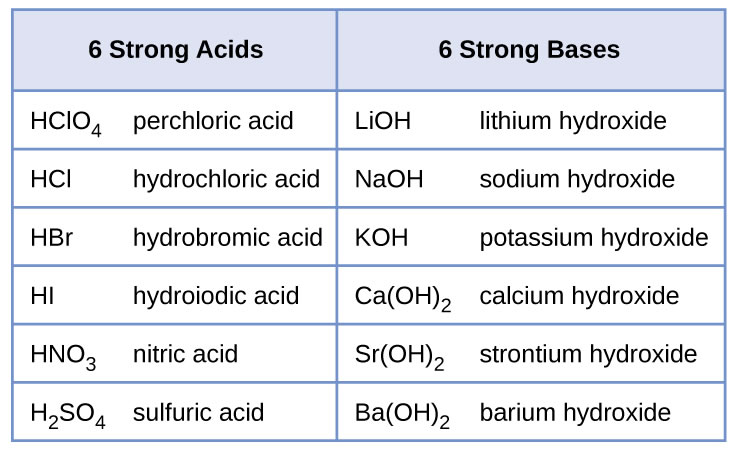
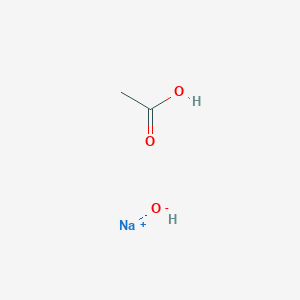
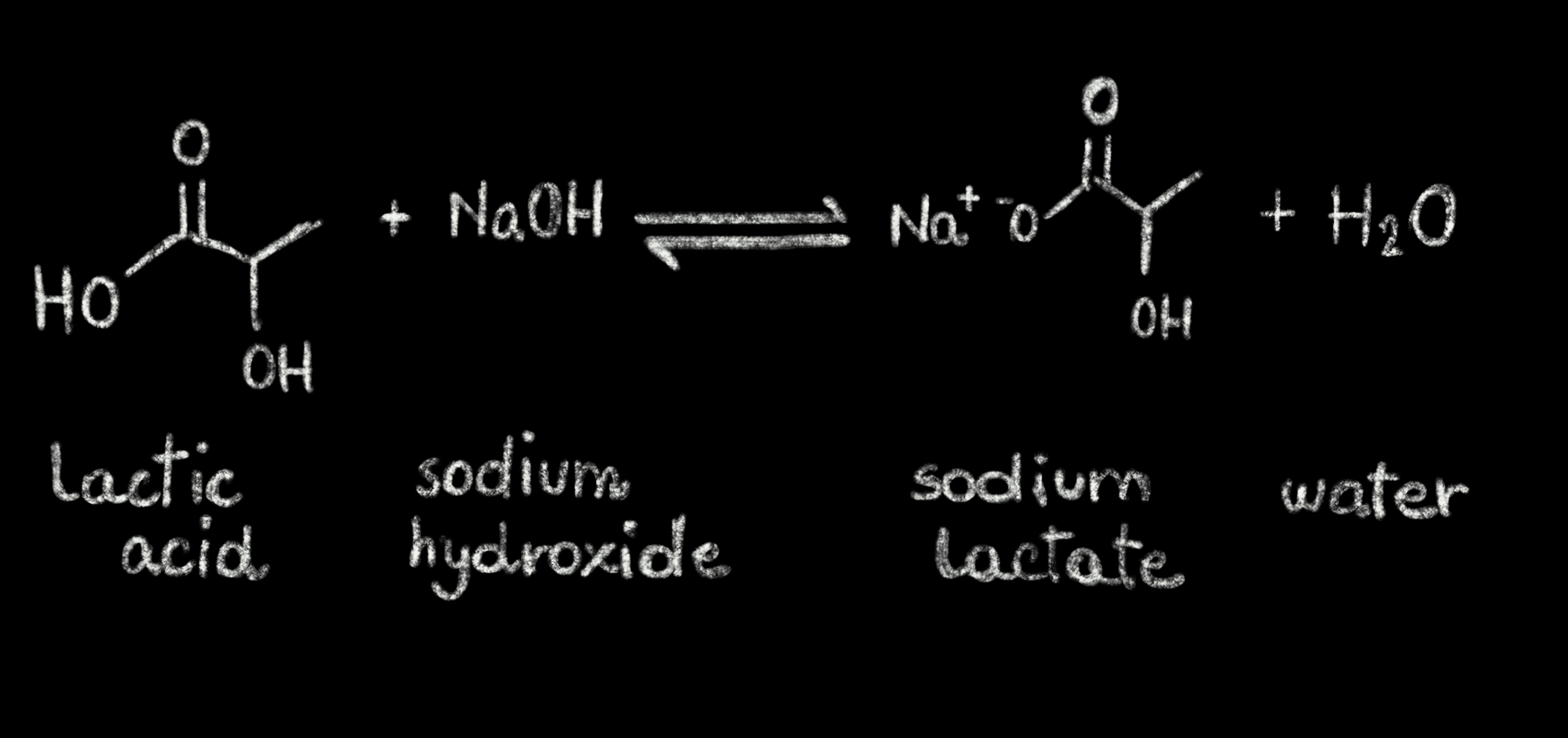
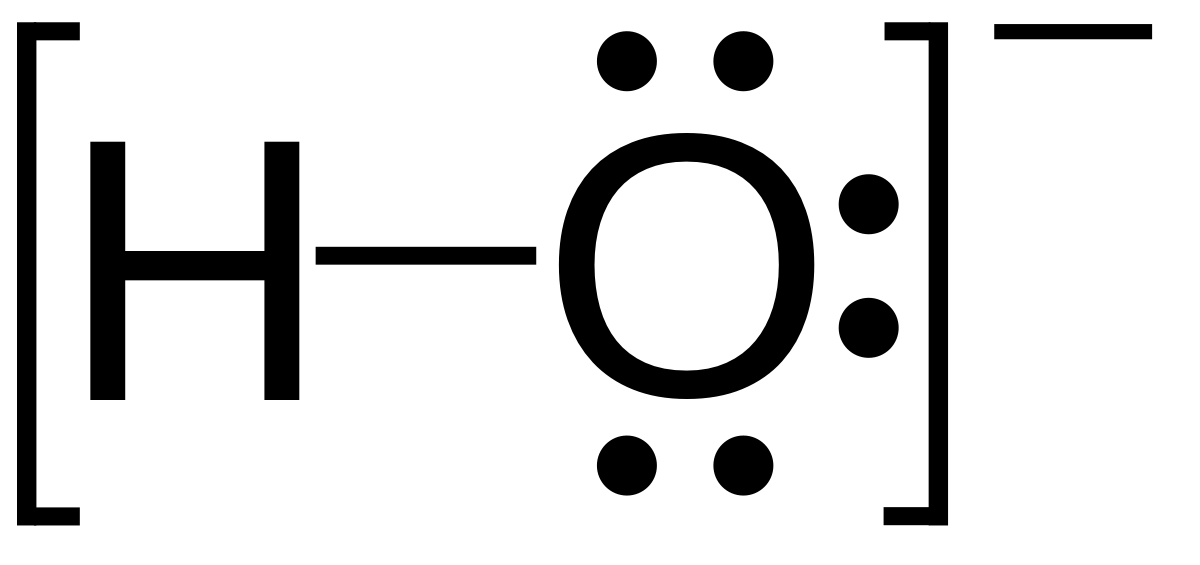
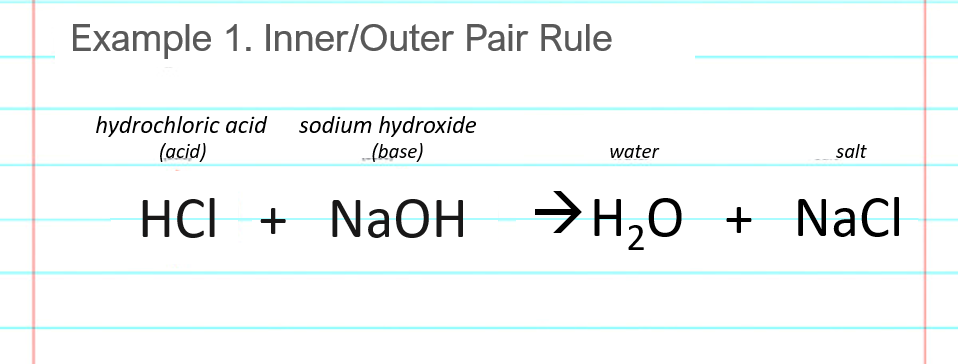
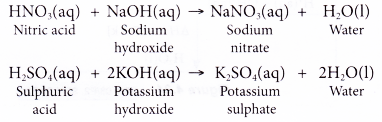Write a balanced chemical equation with state symbols for the reaction : sodium hydroxide solution(in water) reacts with hydrochloric acid solution(in water) to produce sodium chloride and water

Write the neutralization reaction between Hydrochloric acid HCI and sodium hydroxide NaOH, and write the equation for this process.

Acid-base, neutralization reaction of hydrochloric acid and sodium hydroxide. types of chemical reactions, part 6 of 7. | CanStock