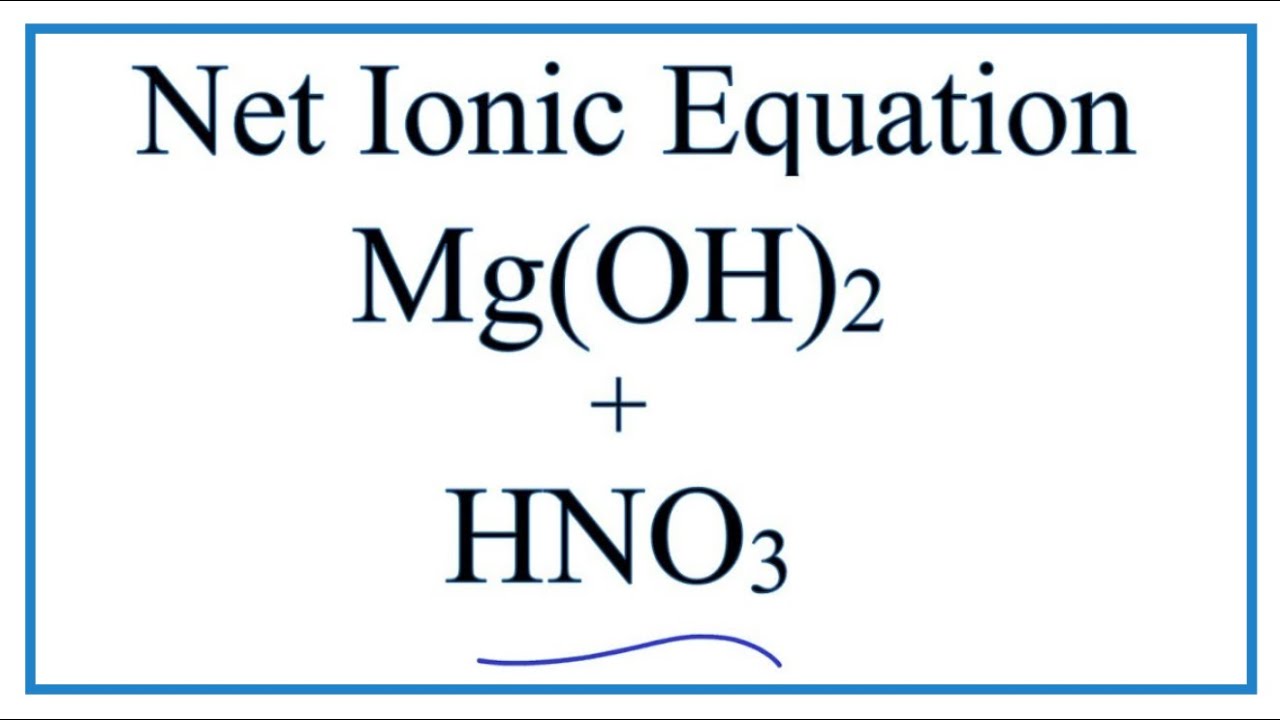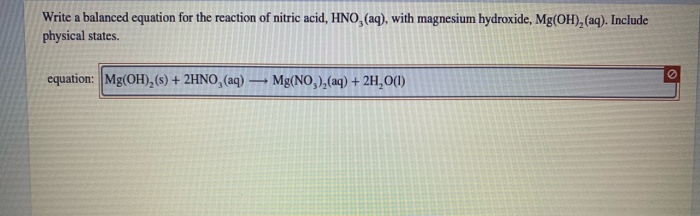SOLVED: 20.A sample of 7.75 g of magnesium hydroxide is added to 25.0 mL of 0.2OOM nitric acid Write balanced equation for the reaction that occurs b. What is the limiting reagent

EP0858985A2 - Aqueous alkaline earth nitrate fertilizer composition and process for making same - Google Patents
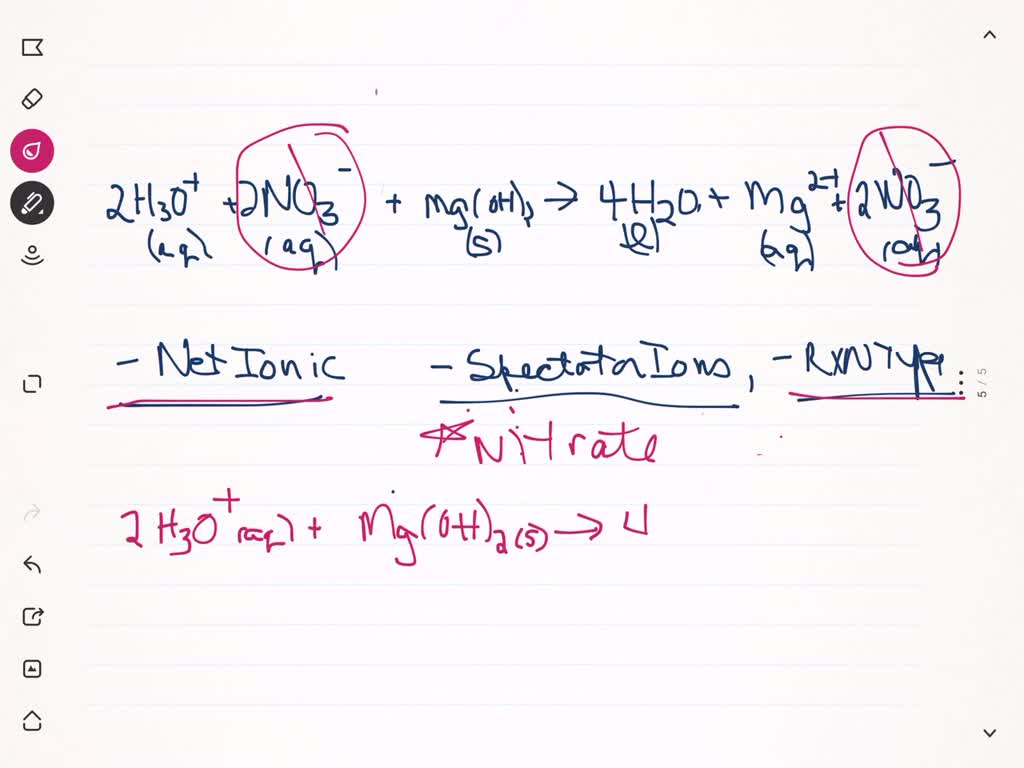
SOLVED:Write the net ionic equation and identify the spectator ion or ions in the reaction of nitric acid and magnesium hydroxide. What type of reaction is this? 2 H3O^+(aq)+2 NO3^-(aq)+Mg(OH)2(s) → 4

Solution For: Write two equations for the neutralization of nitric acid, HNO, with magnesium hydroxide, - Brainly.com

SOLVED: Magnesium nitrate can be produced from the reaction of magnesium hydroxide and nitric acid according to the reaction: MBIOHIz 2 HNOg MB(NOzh 2 Hzo How many grams of MB(NOah can be

Write the balanced chemical equations for the following reaction:Calcium hydroxide + Carbon dioxide → Calcium Carbonate + Water.