Real-Time Studies of Iron Oxalate-Mediated Oxidation of Glycolaldehyde as a Model for Photochemical Aging of Aqueous Tropospheri

Sustainable Iron-Making Using Oxalic Acid: The Concept, A Brief Review of Key Reactions, and An Experimental Demonstration of the Iron-Making Process | ACS Sustainable Chemistry & Engineering
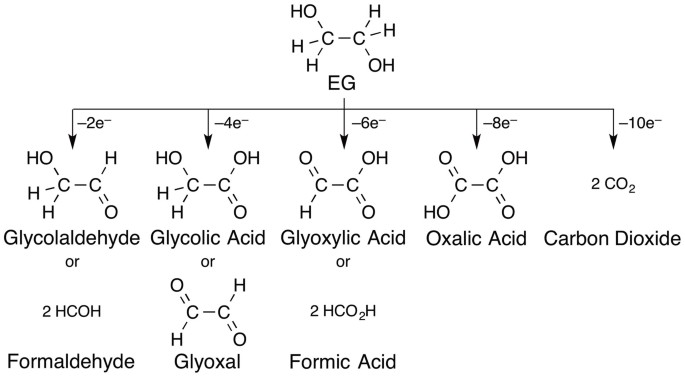
CO2-Free Power Generation on an Iron Group Nanoalloy Catalyst via Selective Oxidation of Ethylene Glycol to Oxalic Acid in Alkaline Media | Scientific Reports
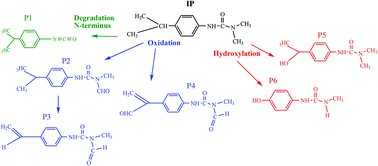
The effect of natural iron oxide and oxalic acid on the photocatalytic degradation of isoproturon: a kinetics and analytical study - Photochemical & Photobiological Sciences (RSC Publishing)

Arsenic mobilization from iron oxides in the presence of oxalic acid under hydrodynamic conditions - ScienceDirect
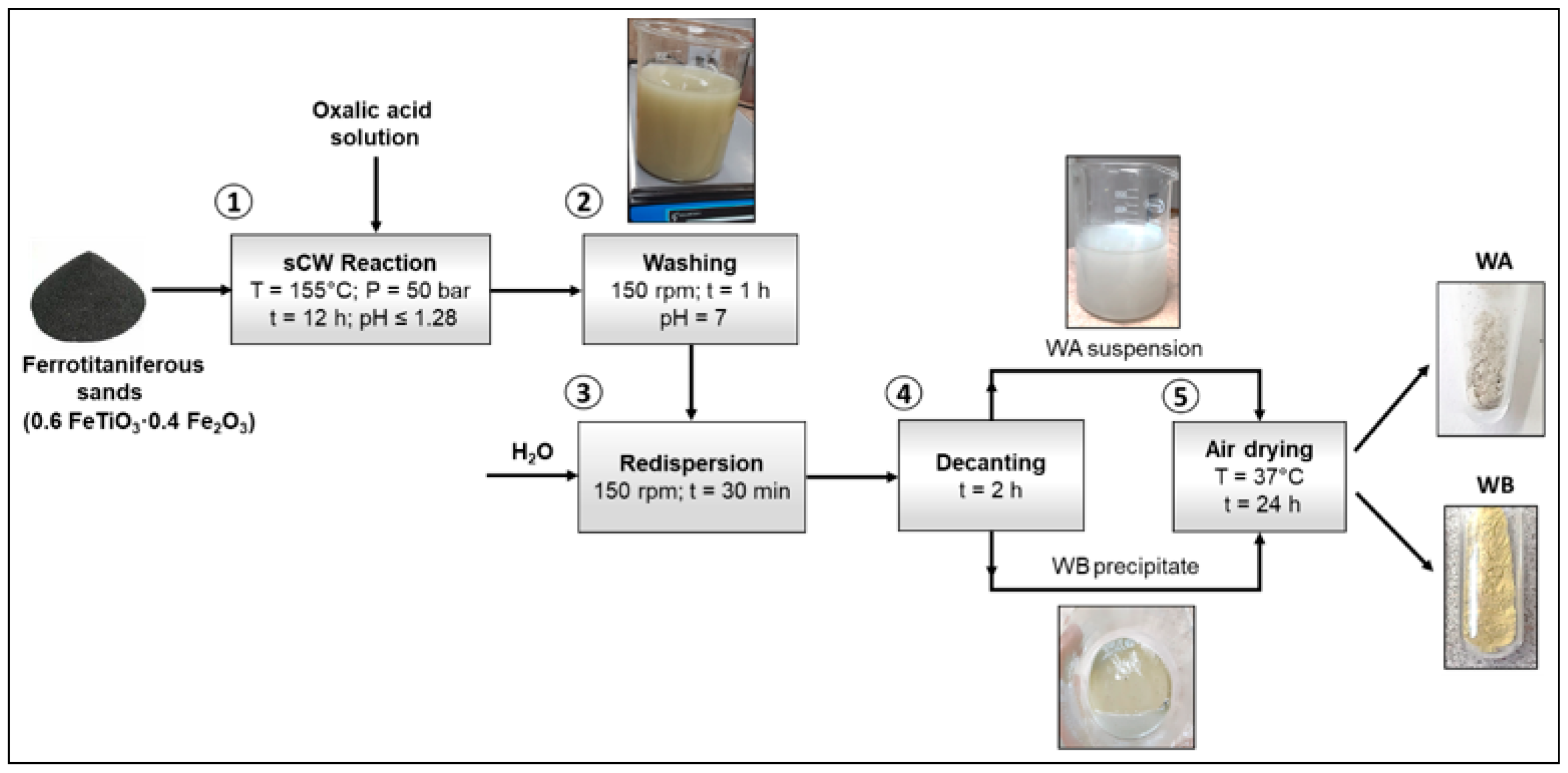
Minerals | Free Full-Text | One-Step Synthesis of Iron and Titanium-Based Compounds Using Black Mineral Sands and Oxalic Acid under Subcritical Water Conditions
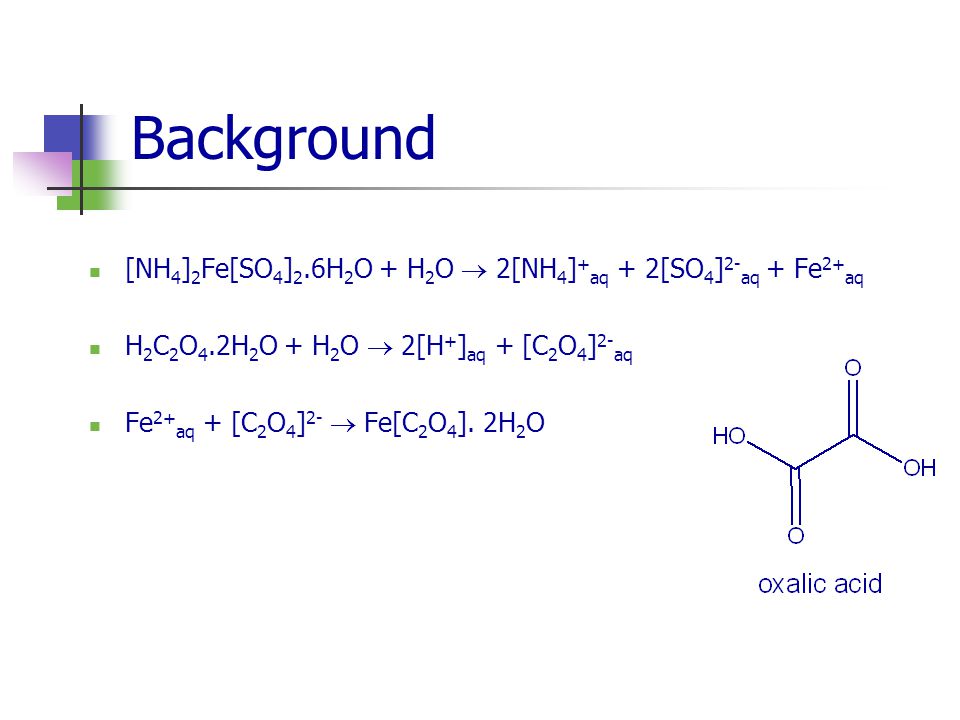
Experiment 5 Synthesis of an Iron Oxalate Compound Chemistry Department UCC1 st Year Practicals. - ppt download

SciELO - Brazil - Thermodynamic analysis of stability in iron removal from kaolin by using oxalic acid Thermodynamic analysis of stability in iron removal from kaolin by using oxalic acid

Sustainable Iron-Making Using Oxalic Acid: The Concept, A Brief Review of Key Reactions, and An Experimental Demonstration of the Iron-Making Process | ACS Sustainable Chemistry & Engineering
The effect of natural iron oxide and oxalic acid on the photocatalytic degradation of isoproturon: a kinetics and analytical study - Photochemical & Photobiological Sciences (RSC Publishing) DOI:10.1039/C6PP00441E

Sustainable Iron-Making Using Oxalic Acid: The Concept, A Brief Review of Key Reactions, and An Experimental Demonstration of the Iron-Making Process | ACS Sustainable Chemistry & Engineering

Oxalic Acid Recovery from High Iron Oxalate Waste Solution by a Combination of Ultrasound-Assisted Conversion and Cooling Crystallization | ACS Sustainable Chemistry & Engineering