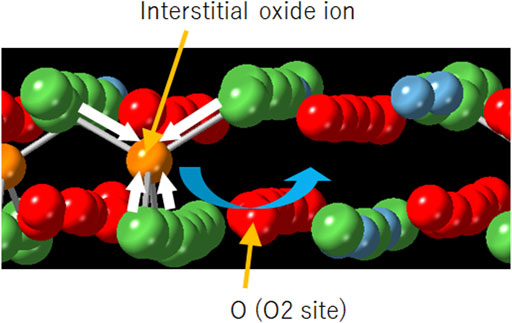
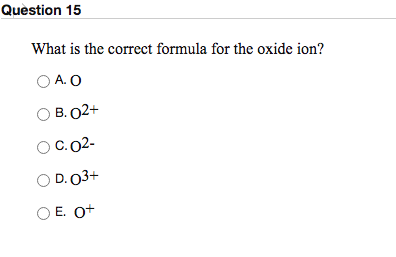Improved oxide-ion conductivity by substitution of Sr for Bi in Dion-Jacobson phase CsBi2Ti2NbO10 - ScienceDirect

Ruddlesden–Popper Oxychlorides Ba3Y2O5Cl2, Sr3Sc2O5Cl2, and Sr2ScO3Cl: First Examples of Oxide-Ion-Conducting Oxychlorides | ACS Applied Energy Materials

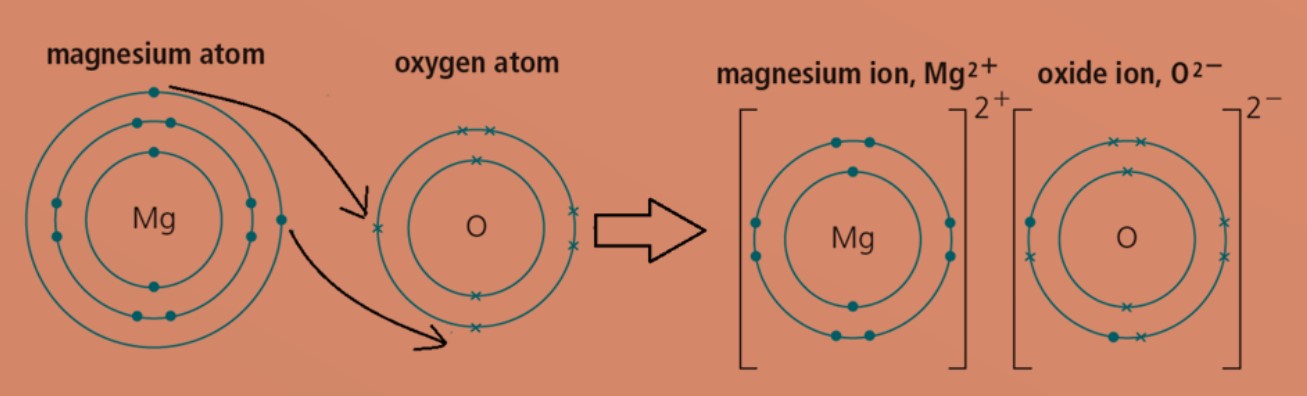
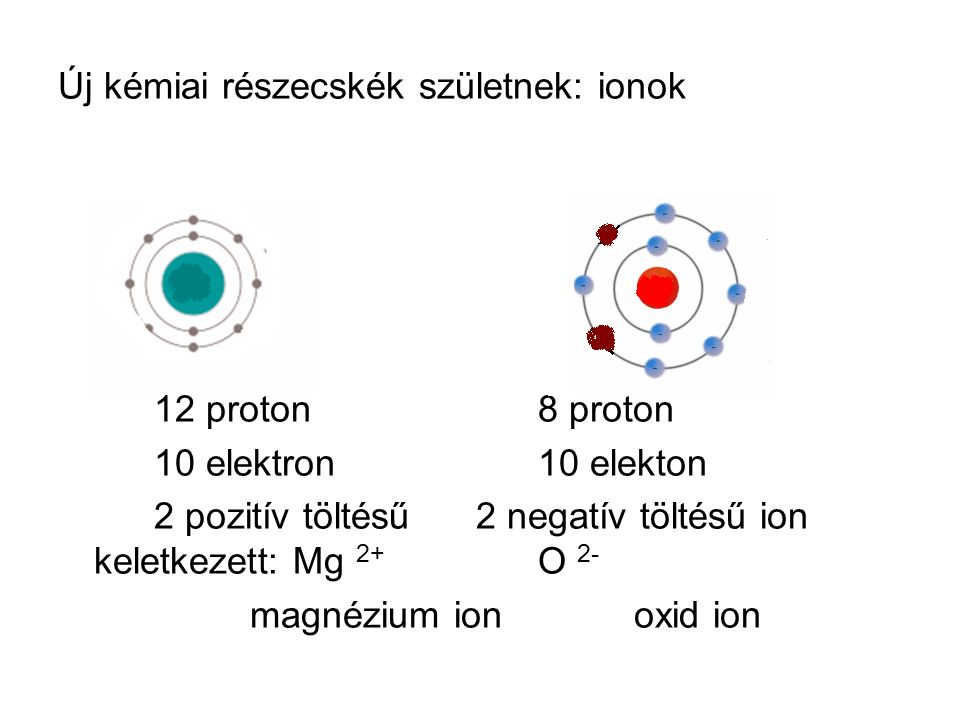
Előadás, mgo, kötés, ábra, oxid, magnézium, jón. Előadás, kötés, ábra, oxid, magnézium, jón. | CanStock

a)(b) The trajectories of oxide-ion conduction processes along the c... | Download Scientific Diagram

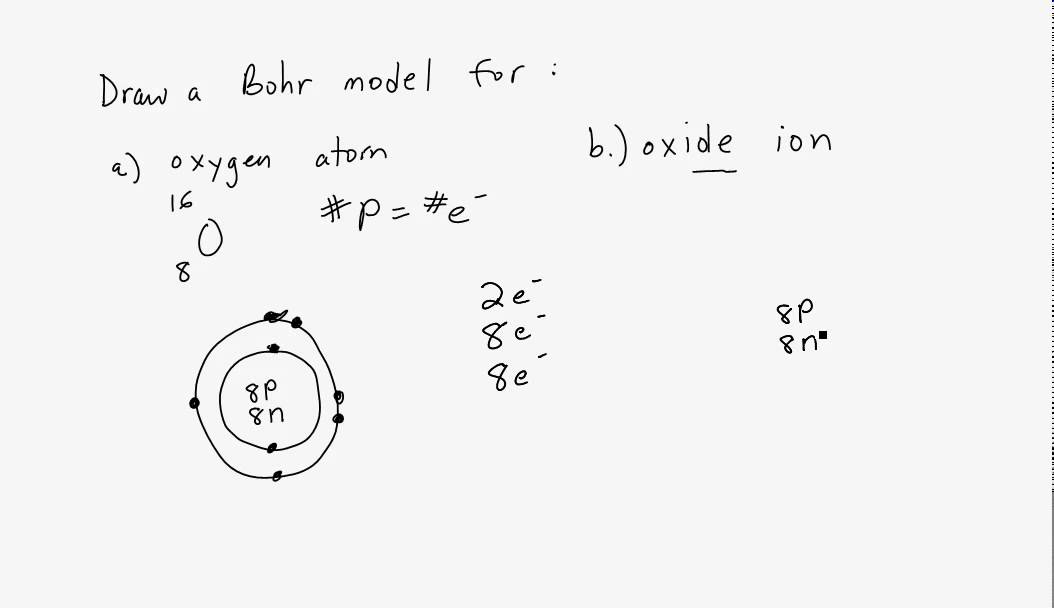
The formation of the oxide ion, O ^2 - (g) , from oxygen atom requires first an exothermic and then an endothermic step as shown below: O(g) + e^-→ O^-(g); Δ H^∘ = -
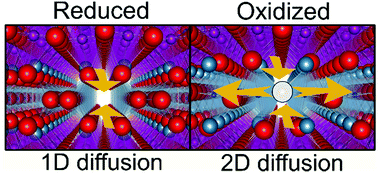
Oxide-ion diffusion in brownmillerite-type Ca2AlMnO5+δ from first-principles calculations - Physical Chemistry Chemical Physics (RSC Publishing)

Chemistry - Chemical Bonding (18 of 35) Lewis Structures for Ionic Comp - Aluminum Oxide - Al2O3 - YouTube

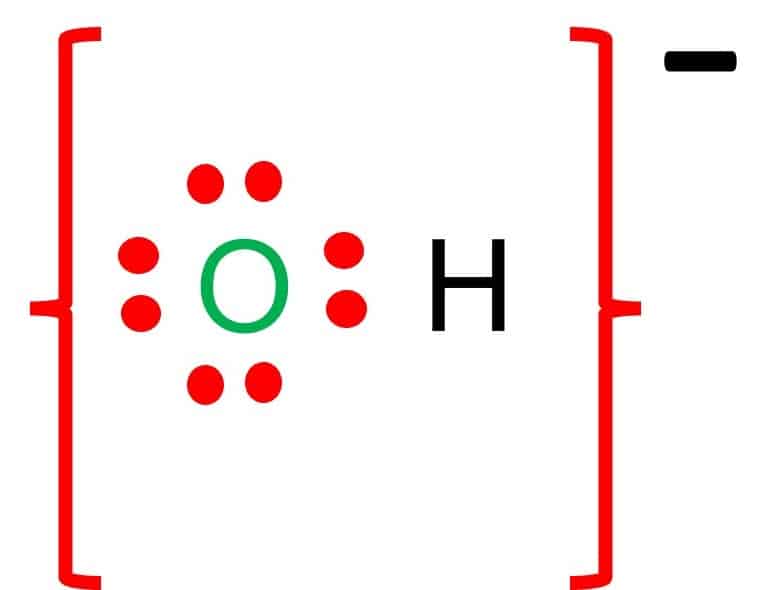
Question Video: Deducing the Ionic Formula of an Ionic Compound Where Both Ions Have Greater-Than-One Charge | Nagwa

Corrosion behavior of oxide ion conductors for high‐temperature direct electrochemical metal oxide reduction - Hwang - 2021 - International Journal of Applied Ceramic Technology - Wiley Online Library

The formation of the oxide ion, O^2 - (g) , from oxygen atom requires first an exothermic and then an endothermic step as shown below: O(g) + e^ - → O^ - (
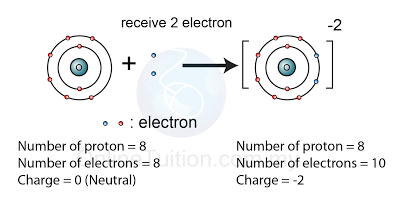
What is the difference between atoms and ions; and covalent compounds and ionic compounds? | Socratic