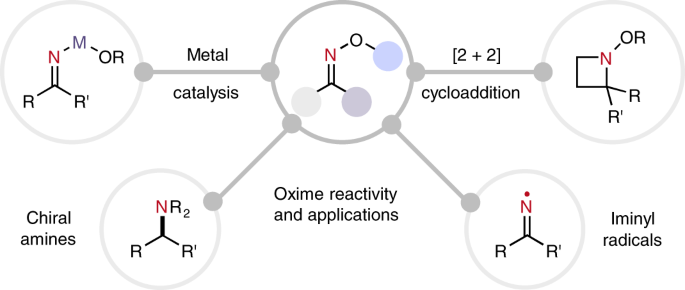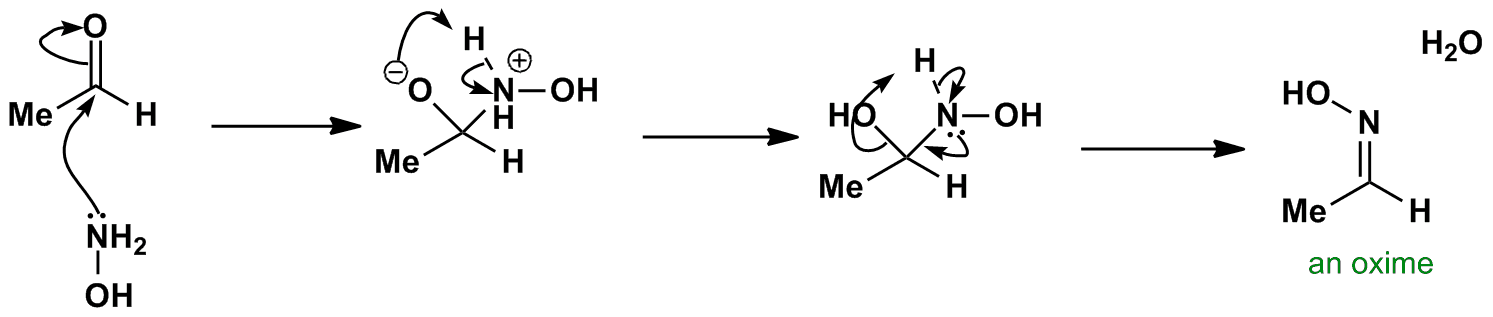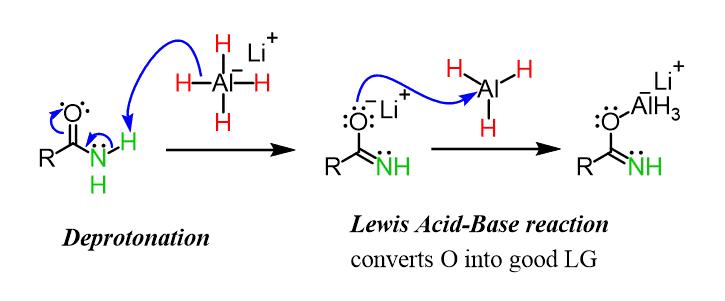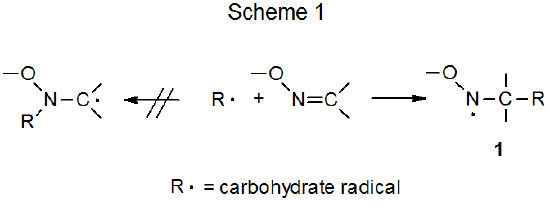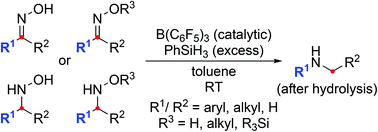
Mild reductive rearrangement of oximes and oxime ethers to secondary amines with hydrosilanes catalyzed by B(C6F5)3 - Organic Chemistry Frontiers (RSC Publishing)
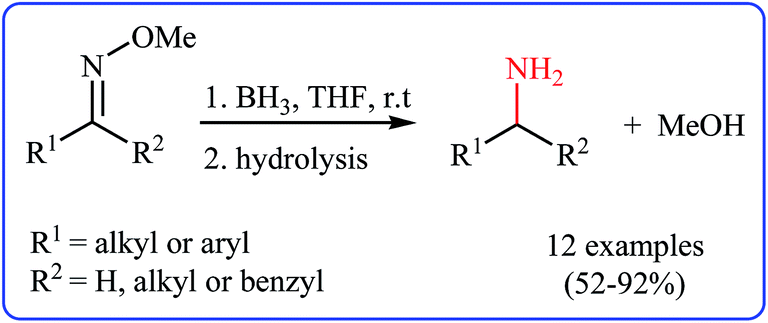
Oxime ethers as versatile precursors in organic synthesis: a review - RSC Advances (RSC Publishing) DOI:10.1039/C5RA15299B

The local and natural sources in synthetic methods: the practical synthesis of aryl oximes from aryl aldehydes under catalyst-free conditions in mineral water | SpringerLink
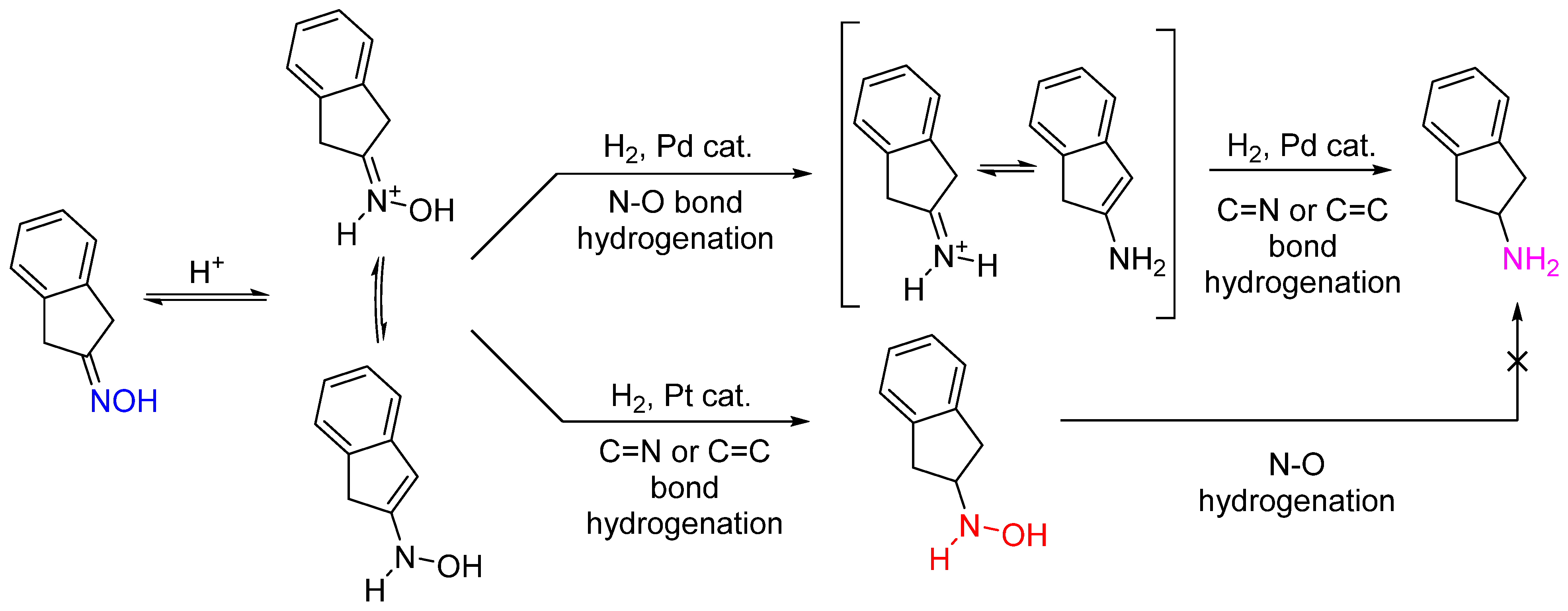
Catalysts | Free Full-Text | Heterogeneous Catalysis as an Efficient Tool for Selective Hydrogenation of Oximes to Amines and Hydroxylamines

Use your understanding of organic chemistry to provide the mechanism and the reagents for the three-step transformation that gives the following product (* is the radio labeled carbon 14C). | Homework.Study.com

Preparation of amine by reduction of oxime, preparation of amine, neet chemistry, by surendra, - YouTube

Asymmetric Reduction of Oxime Ethers Promoted by Chiral Spiroborate Esters with an O3BN Framework | The Journal of Organic Chemistry


![Reduction of Oximes to Amines Using a Zinc-Copper Couple - [www.rhodium.ws] Reduction of Oximes to Amines Using a Zinc-Copper Couple - [www.rhodium.ws]](https://erowid.org/archive/rhodium/chemistry/pictures/oxime2amine.zn-cu.gif)