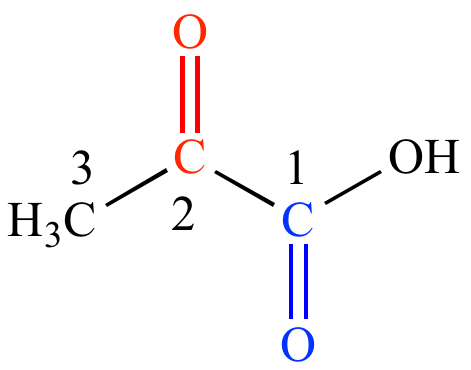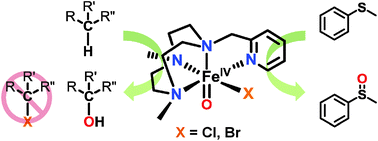
Structural modeling of iron halogenases: synthesis and reactivity of halide-iron(iv)-oxo compounds - Chemical Communications (RSC Publishing)
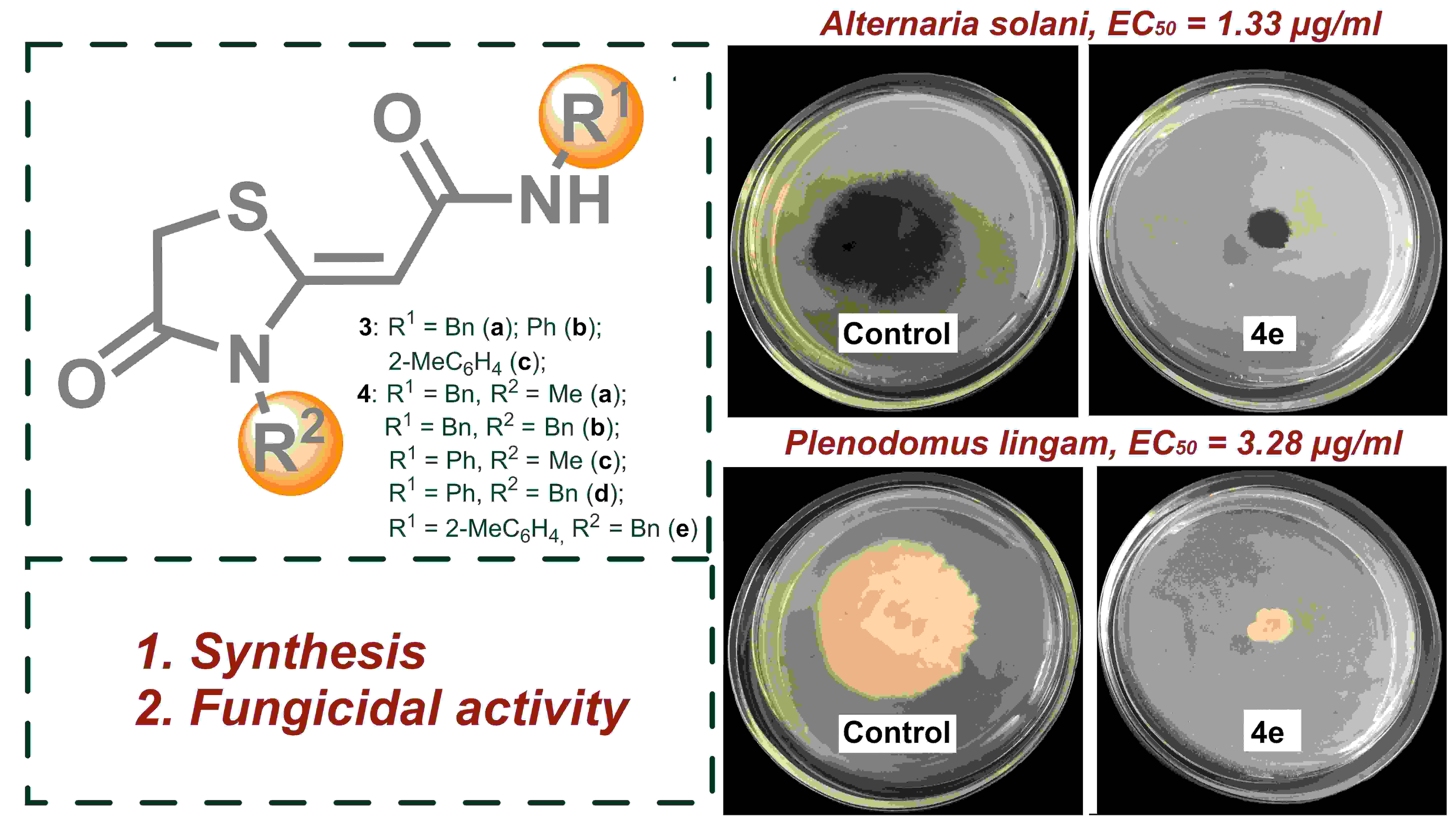
2-(4-Oxo-1,3-thiazolidin-2-ylidene)acetamid as promising scaffold for designing new antifungal compounds | Obydennov | Chimica Techno Acta

Modeling Heme Peroxidase: Heme Saddling Facilitates Reactions with Hyperperoxides To Form High‐Valent FeIV‐Oxo Species - Wu - 2022 - Chemistry – A European Journal - Wiley Online Library


![3-oxo-3-[1-(propan-2-ylamino)cyclobutyl]propanal | Chemical Details | ChemRTP 3-oxo-3-[1-(propan-2-ylamino)cyclobutyl]propanal | Chemical Details | ChemRTP](http://www.chemrtp.com/2d-structure/JFWWGMWUNATSIR-UHFFFAOYSA-N.png)