![Regio- and stereoselectivity of the 1,3-dipolar cycloaddition of azomethine ylides to 3-nitro-2(1H)-quinolinone derivatives: A new synthesis of Pyrrolo[3,4-c] quinolines - ScienceDirect Regio- and stereoselectivity of the 1,3-dipolar cycloaddition of azomethine ylides to 3-nitro-2(1H)-quinolinone derivatives: A new synthesis of Pyrrolo[3,4-c] quinolines - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040402022007499-ga1.jpg)
Regio- and stereoselectivity of the 1,3-dipolar cycloaddition of azomethine ylides to 3-nitro-2(1H)-quinolinone derivatives: A new synthesis of Pyrrolo[3,4-c] quinolines - ScienceDirect

András DANCSÓ | Senior Researcher | Dr. techn. | Laboratory of Structural Chemistry | Research profile

Propylphosphonic anhydride (T3P®) mediated one-pot three-component synthesis of racemic dialkyl (2-substituted-3-oxo-2,3-dihydro-1H-isoindol-1-yl)phosphonates - ScienceDirect
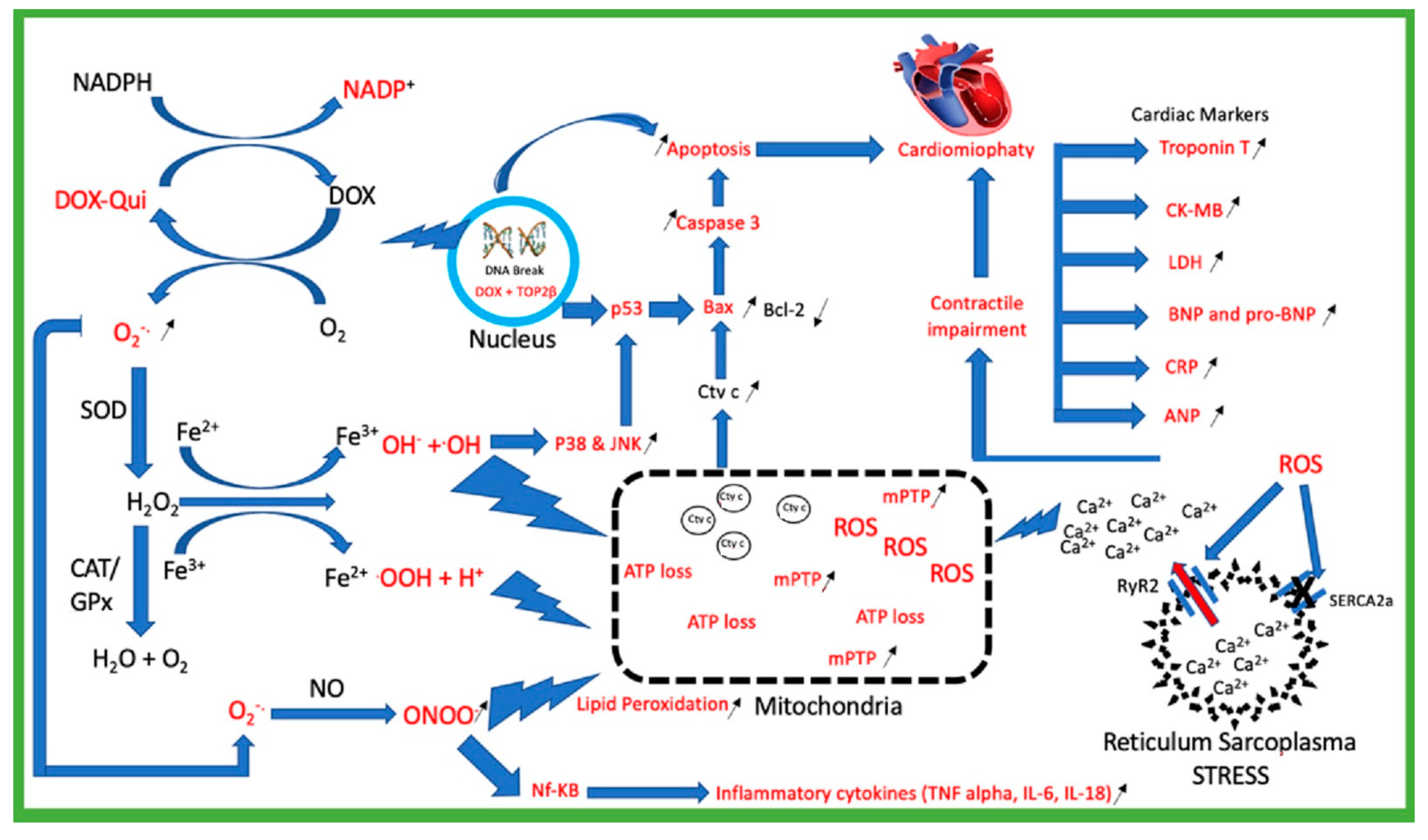
Molecules | Free Full-Text | The Role of Flavonoids as a Cardioprotective Strategy against Doxorubicin-Induced Cardiotoxicity: A Review
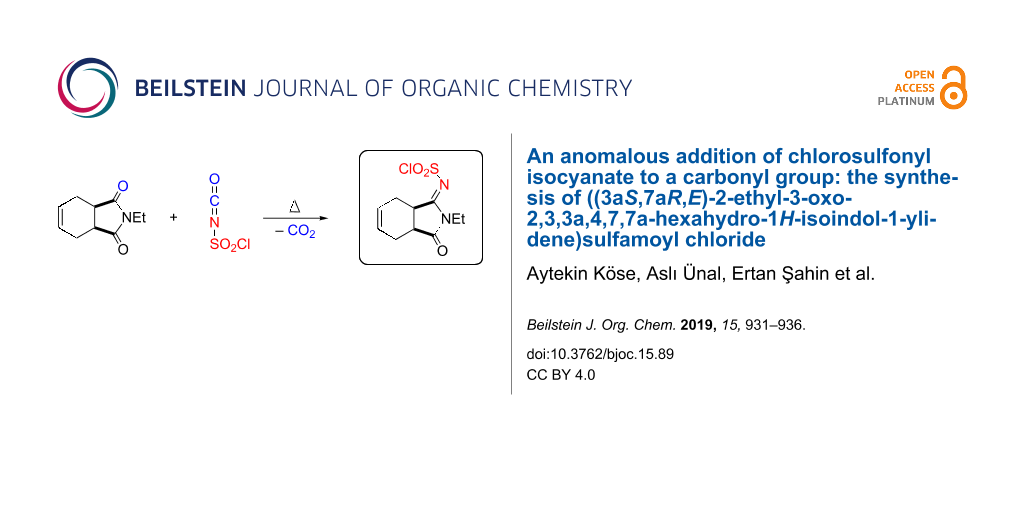
BJOC - An anomalous addition of chlorosulfonyl isocyanate to a carbonyl group: the synthesis of ((3aS,7aR,E)-2-ethyl-3-oxo-2,3,3a,4,7,7a-hexahydro-1H-isoindol-1-ylidene)sulfamoyl chloride

The Use of Copper and Vanadium Mineral Ores in Catalyzed Mechanochemical Carbon–Carbon Bond Formations | ACS Sustainable Chemistry & Engineering
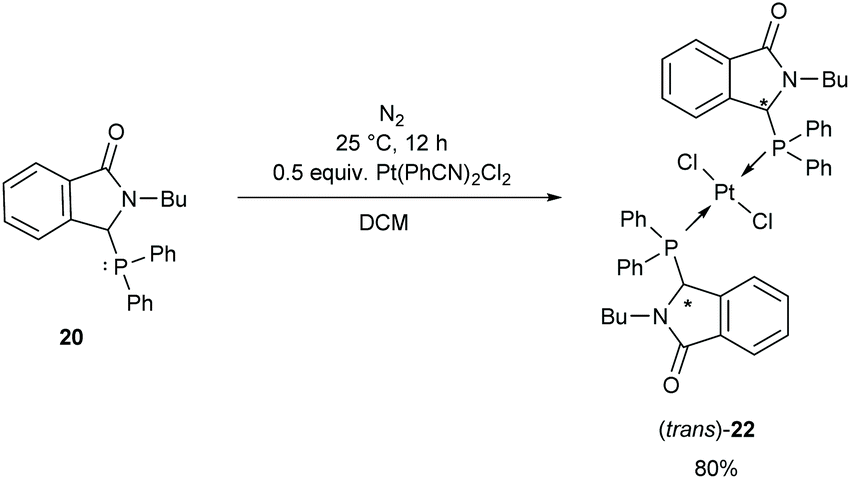
Three-component synthesis, utilization and biological activity of phosphinoyl-functionalized isoindolinones - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D1OB01610E

PDF) Remarkably Efficient Synthesis of 2 H -Indazole 1-Oxides and 2 H -Indazoles via Tandem Carbon−Carbon Followed by Nitrogen−Nitrogen Bond Formation | Jaroslav Zajícek - Academia.edu

The Ugi three-component reaction and its variants - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00313E

Propylphosphonic anhydride (T3P®) mediated one-pot three-component synthesis of racemic dialkyl (2-substituted-3-oxo-2,3-dihydro-1H-isoindol-1-yl)phosphonates - ScienceDirect















