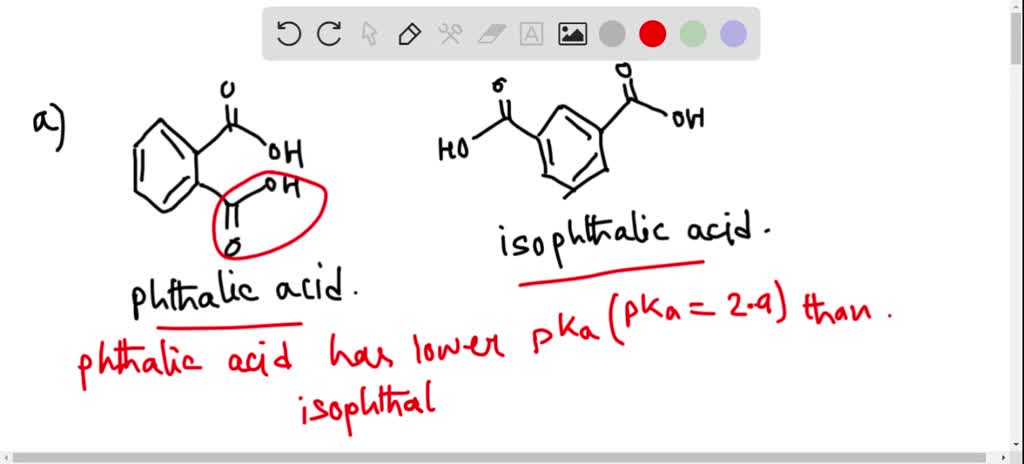
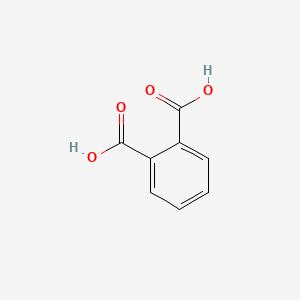
SOLVED:Phthalic acid and isophthalic acid have protons on two carboxy groups that can be removed with base. (a) Explain why the p Ka for loss of the first proton (p Ka 1)
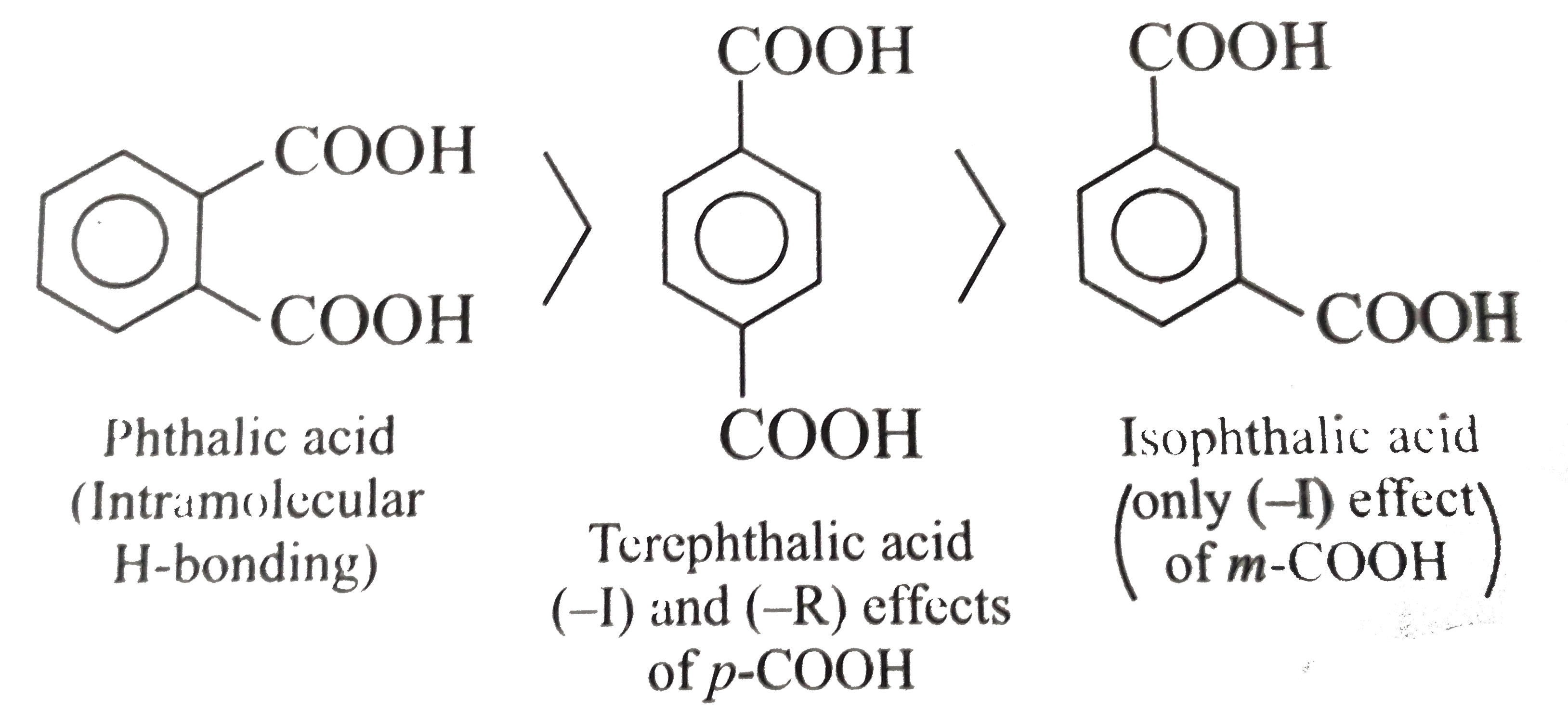
Explain the following : (a) Highly branched acids such as are less acidic than unbranched acids. (b) K(a1) for maleic acid is greater than that for fumaric acid or pK(a1) for maleic
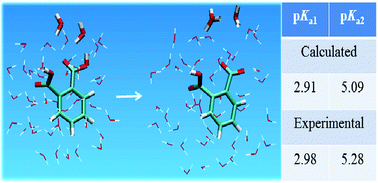
Estimating successive pKa values of polyprotic acids from ab initio molecular dynamics using metadynamics: the dissociation of phthalic acid and its isomers - Physical Chemistry Chemical Physics (RSC Publishing)

A Reliable and Efficient First Principles-Based Method for Predicting pKa Values. 2. Organic Acids | The Journal of Physical Chemistry A

Enhanced Solubility of Telmisartan Phthalic Acid Cocrystals within the pH Range of a Systemic Absorption Site | ACS Omega



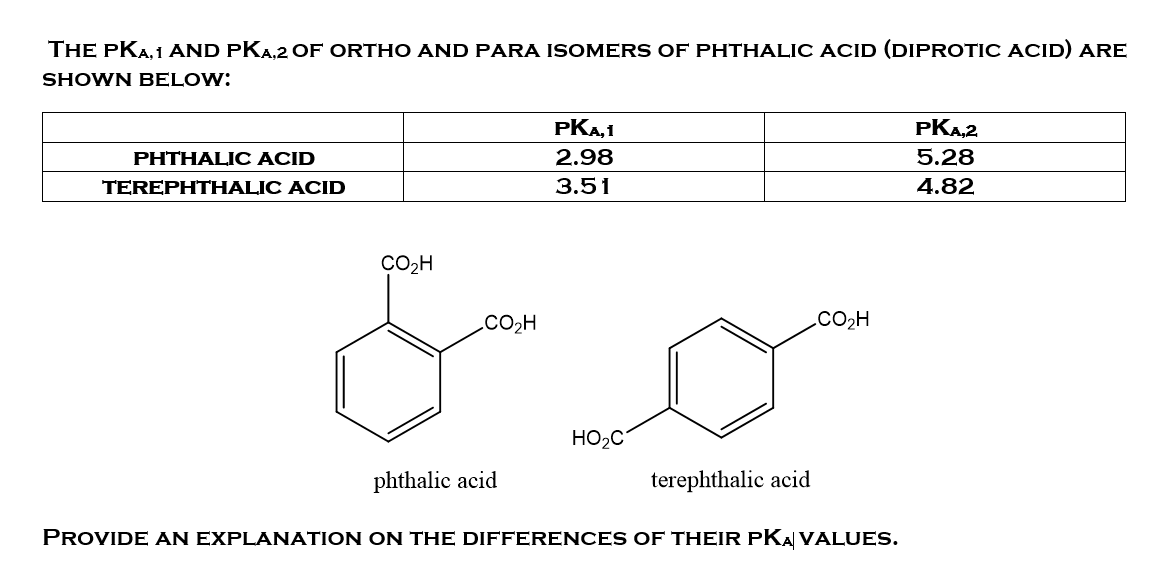
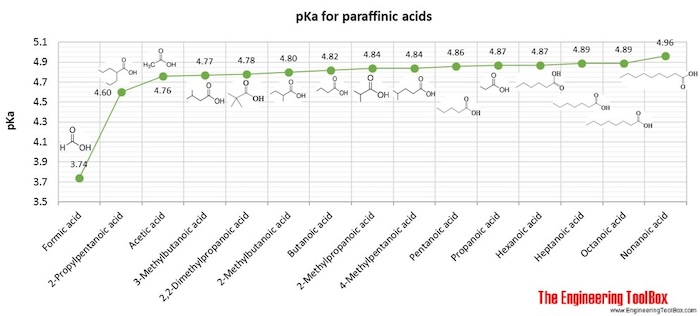




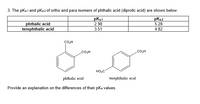

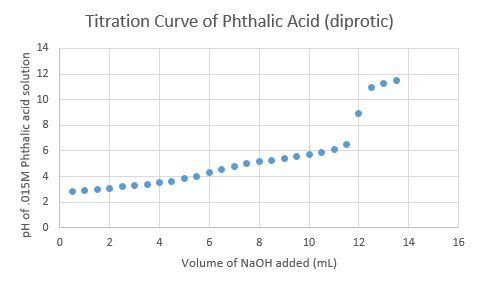
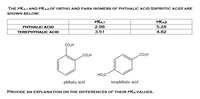


![PDF] Trends in pKa Values for polyprotic carboxylic acids | Semantic Scholar PDF] Trends in pKa Values for polyprotic carboxylic acids | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/cee8fc572e1a28f3ff3aa09345e4884583c46862/8-Table2-1.png)