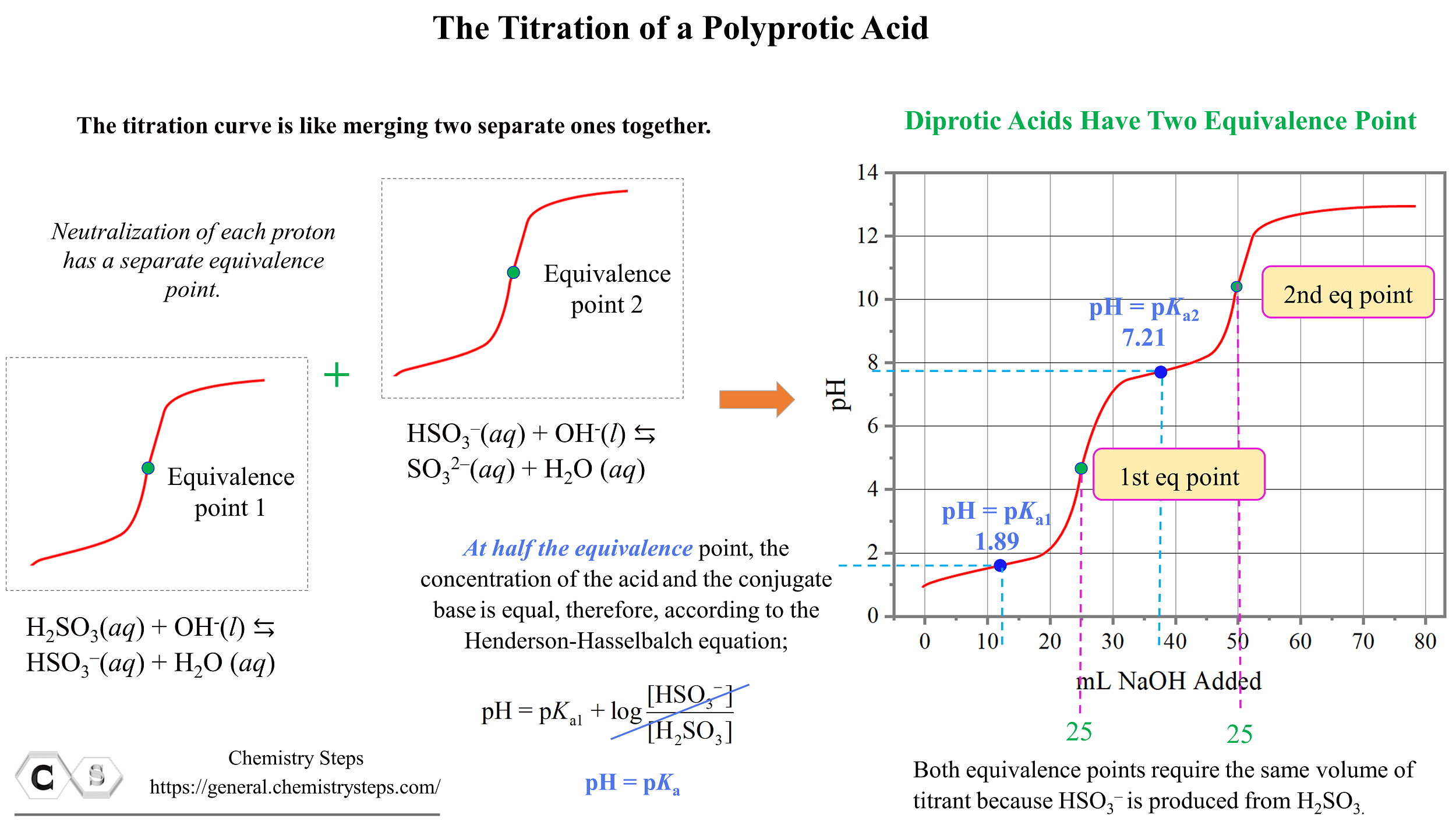
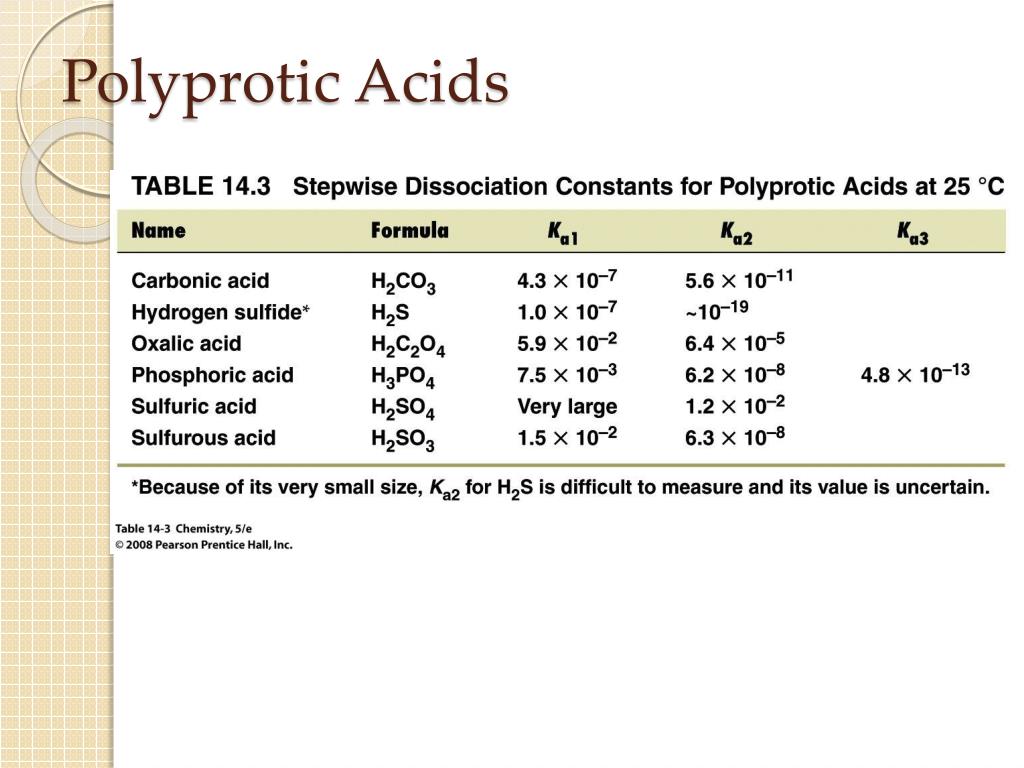
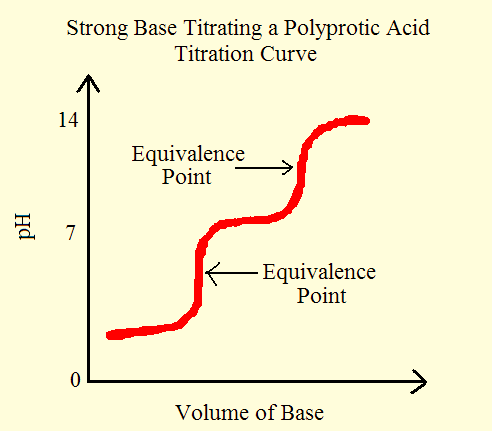
Polyprotic Acid: Introduction, Examples, Demonstration and Dissociation Constant value of different Polyprotic Acid

Table 2 from Cytotoxic effects of polybasic acids, poly(alkenoic acid)s, and the monomers with various functional groups on human pulp fibroblasts. | Semantic Scholar

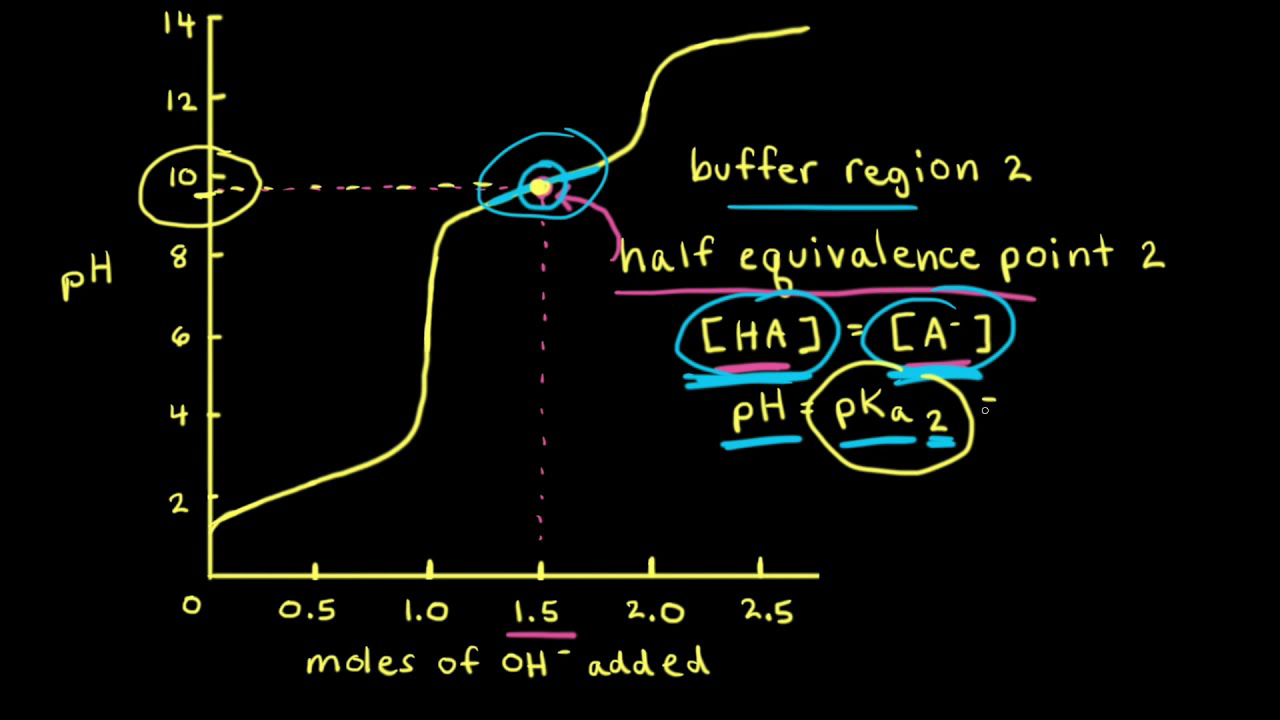
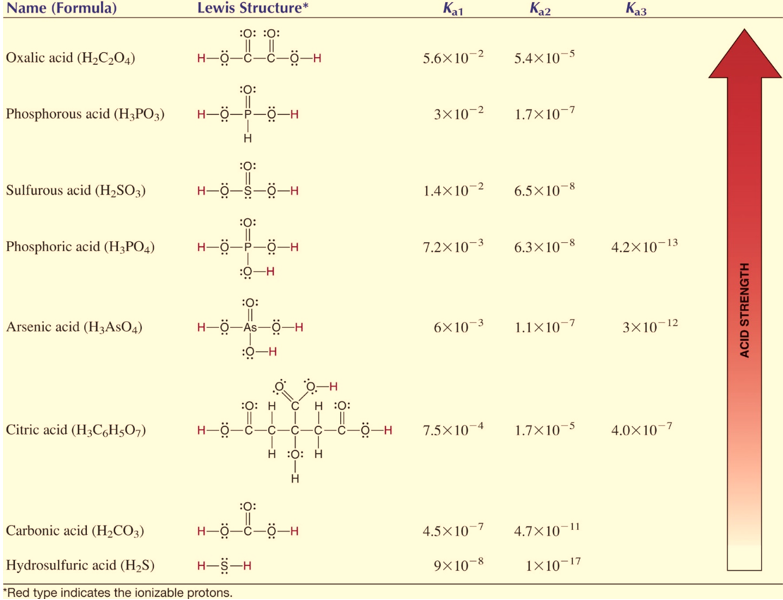
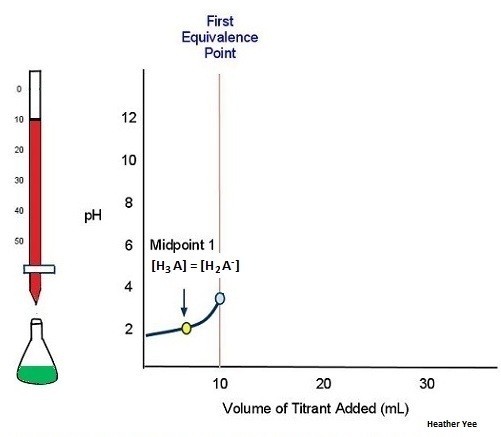
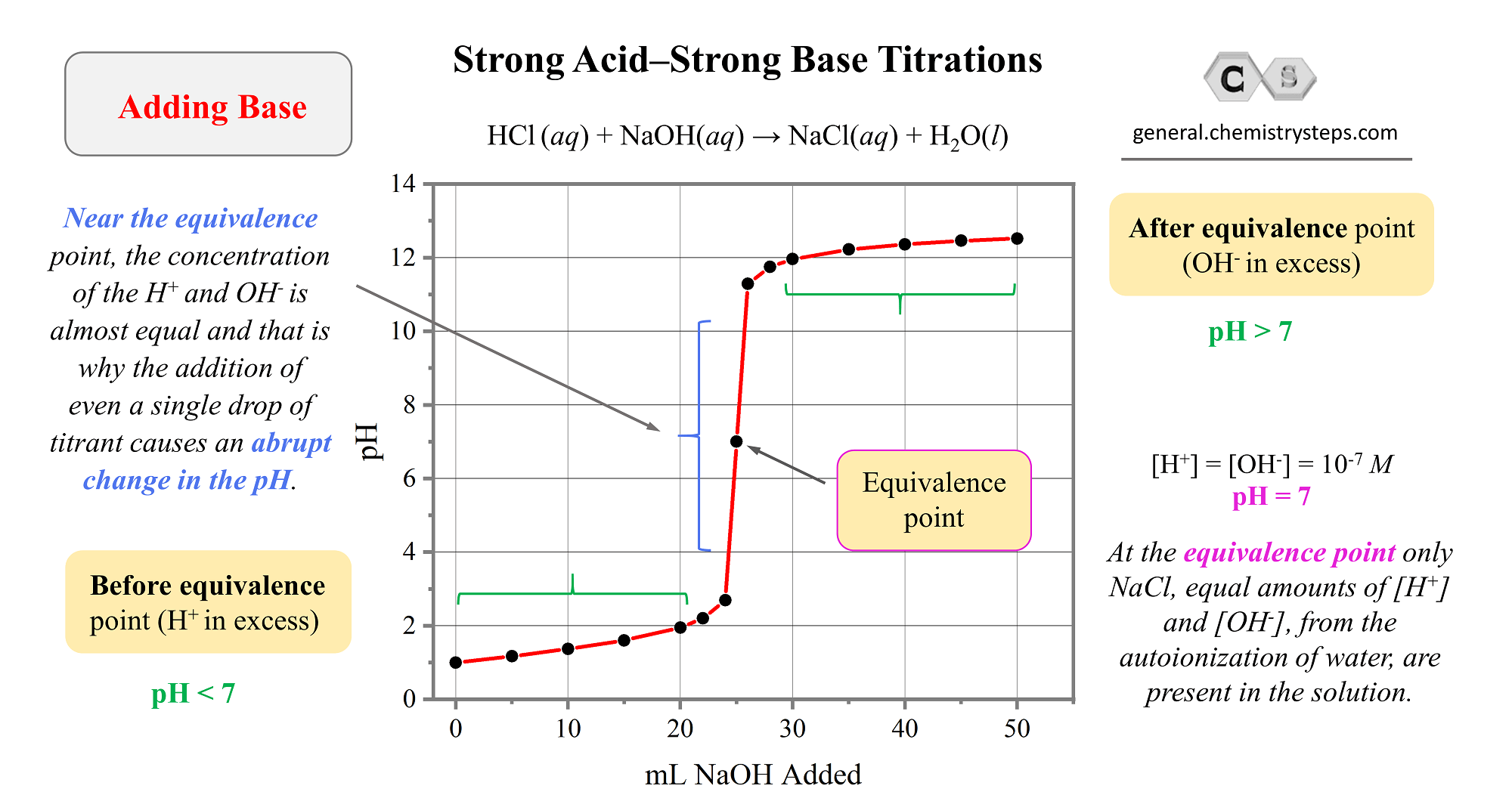
For a polybasic acid, the dissociation constants have different values for each step, e.g., H3A H^+ + H2A^- ; K = Ka1 H2A^- H^+ + HA^2 - ; K = Ka2 HA^2 -