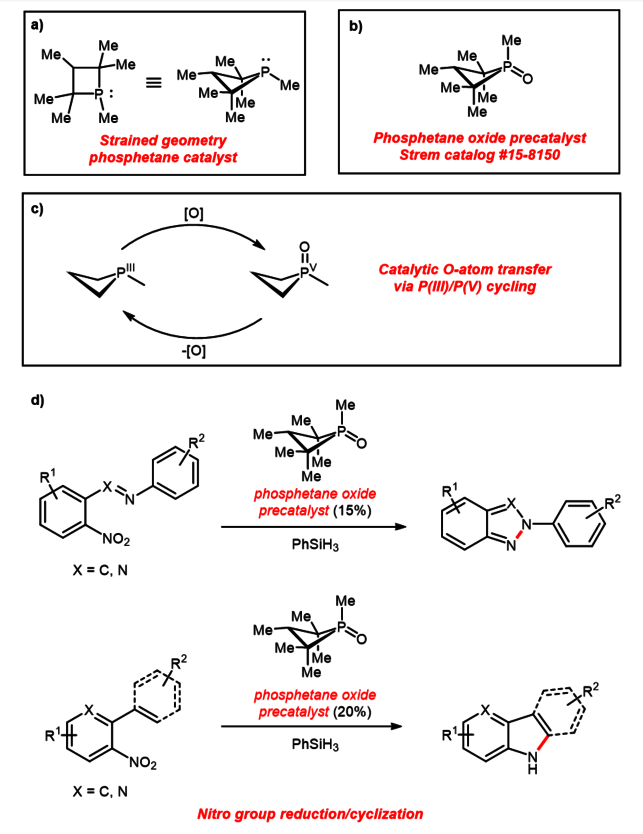
Translate the following statement into chemical equations and then balance the equations: (i) Phosphorus burns in oxygen to give phosphorus pentoxide. (ii) Aluminium metal replaces iron from ferric oxide, Fe(2)O(3), giving aluminium

Oxygen and oxides 2.16 recall the gases present in air and their approximate percentage by volume - ppt video online download
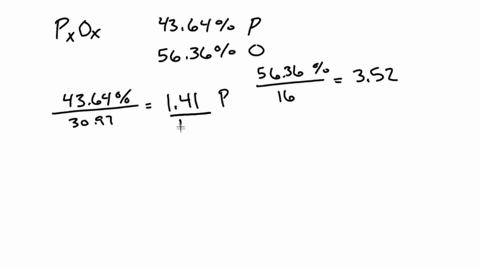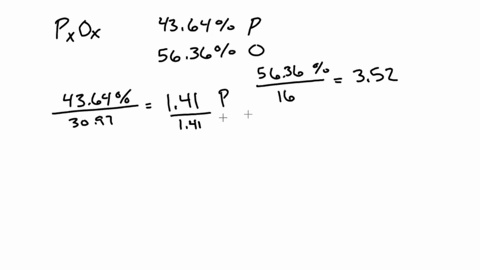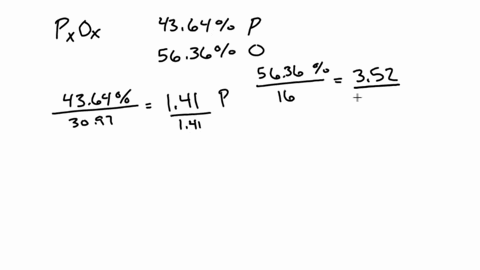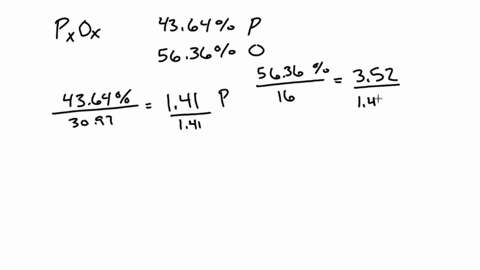
SOLVED:A 1.45-g sample of phosphorus burns in air and forms 2.57 g of a phosphorus oxide. Calculate the empirical formula of the oxide.

Q12 Describe the simple experiments to show the presence of a Oxygen and nitrogen component in air u...
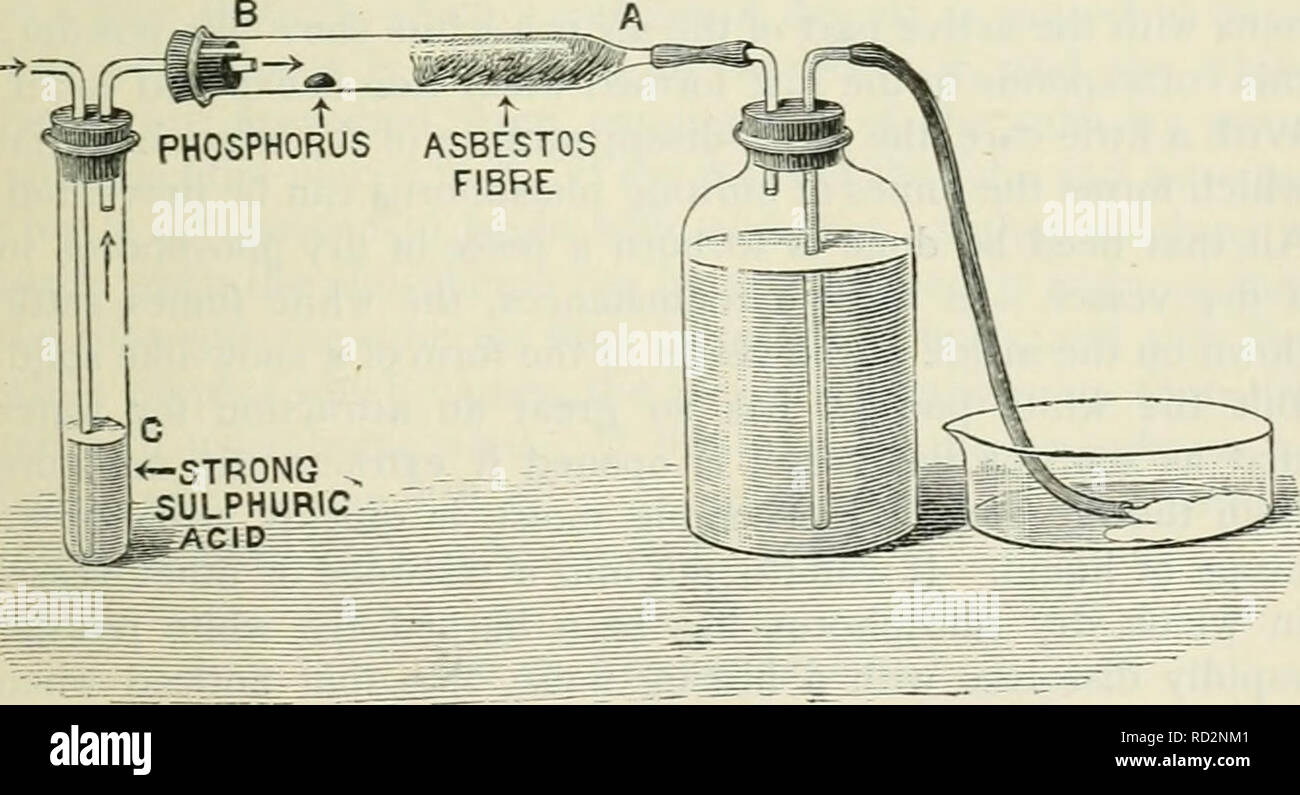
Elementary physics and chemistry: second stage. Science. BURNING PHOSPHORUS IN AIR. 109 powder is deposited upon the sides of the cylinder. When the phosphorus has ceased to burn, hft up the

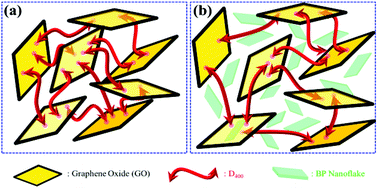
Enhancing thermal oxidation and fire resistance of reduced graphene oxide by phosphorus and nitrogen co-doping: Mechanism and kinetic analysis - ScienceDirect
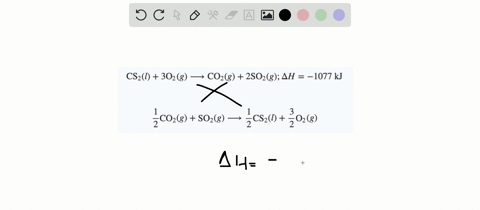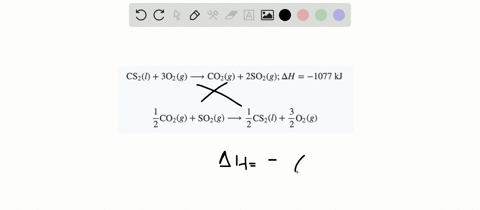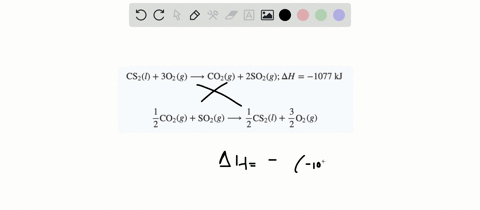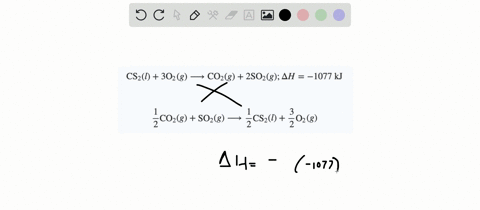
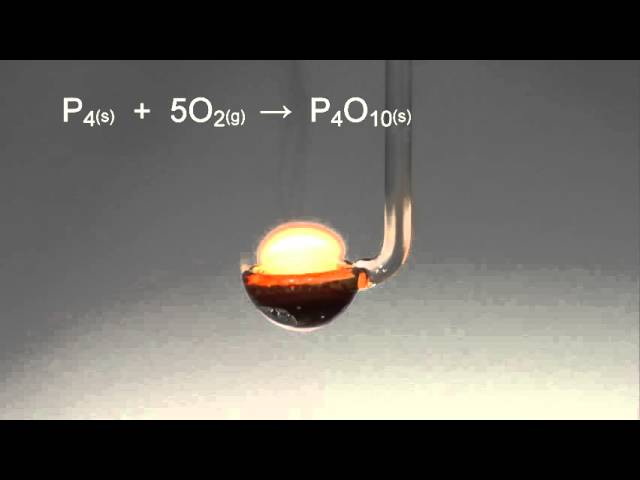
SOLVED:When white phosphorus burns in air, it produces phosphorus(V) oxide. P4(s)+5 O2(g) ⟶P4 O10(s) ; ΔH=-3010 kJ What is ΔH for the following equation? P4 O10(s) ⟶P4(s)+5 O2(g)
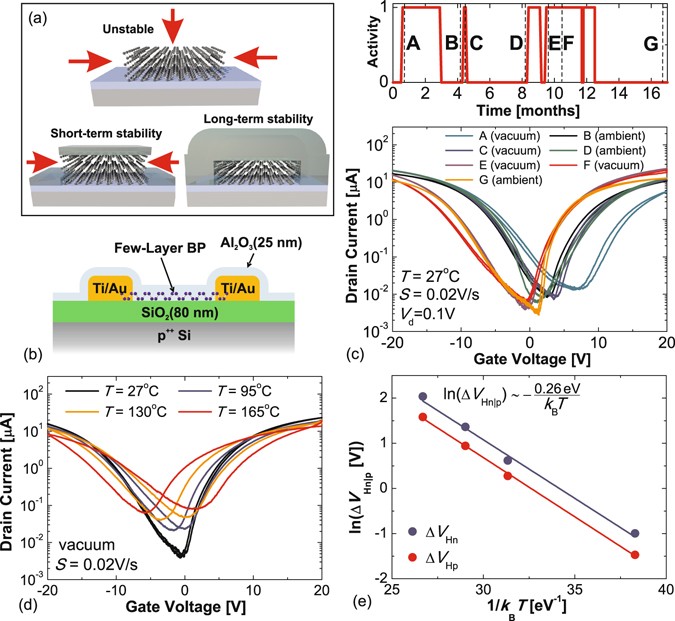
Highly-stable black phosphorus field-effect transistors with low density of oxide traps | npj 2D Materials and Applications
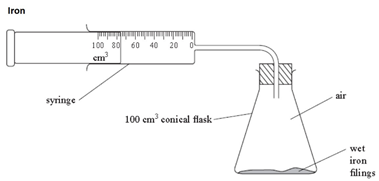
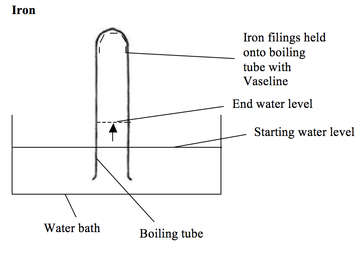
2:10 understand how to determine the percentage by volume of oxygen in air using experiments involving the reactions of metals (e.g. iron) and non-metals (e.g. phosphorus) with air - TutorMyself Chemistry
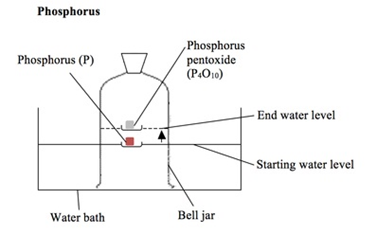
2:10 understand how to determine the percentage by volume of oxygen in air using experiments involving the reactions of metals (e.g. iron) and non-metals (e.g. phosphorus) with air - TutorMyself Chemistry
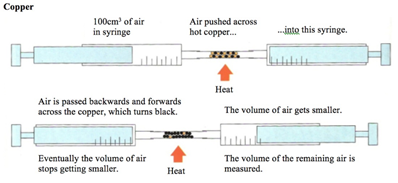
2:10 understand how to determine the percentage by volume of oxygen in air using experiments involving the reactions of metals (e.g. iron) and non-metals (e.g. phosphorus) with air - TutorMyself Chemistry