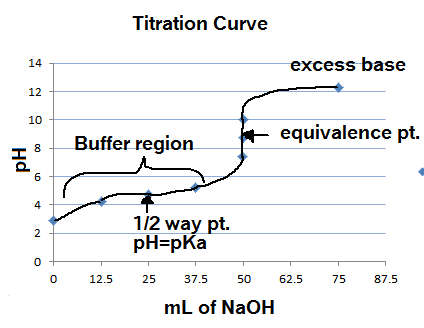
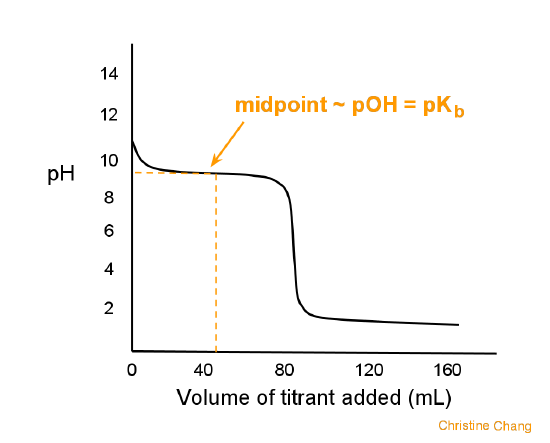
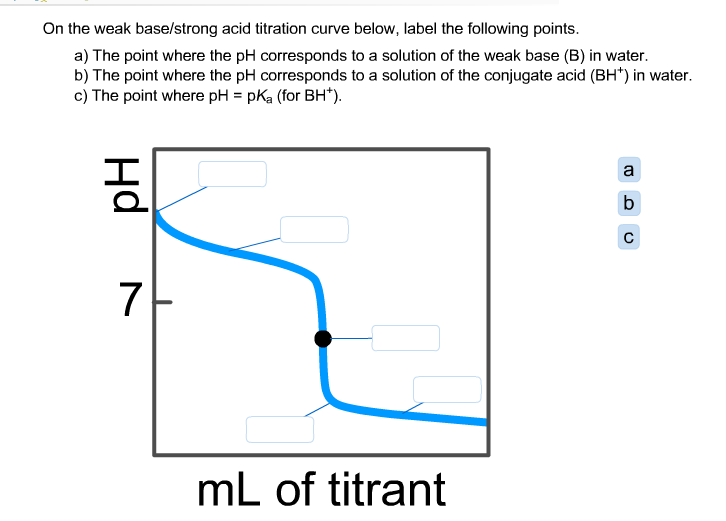
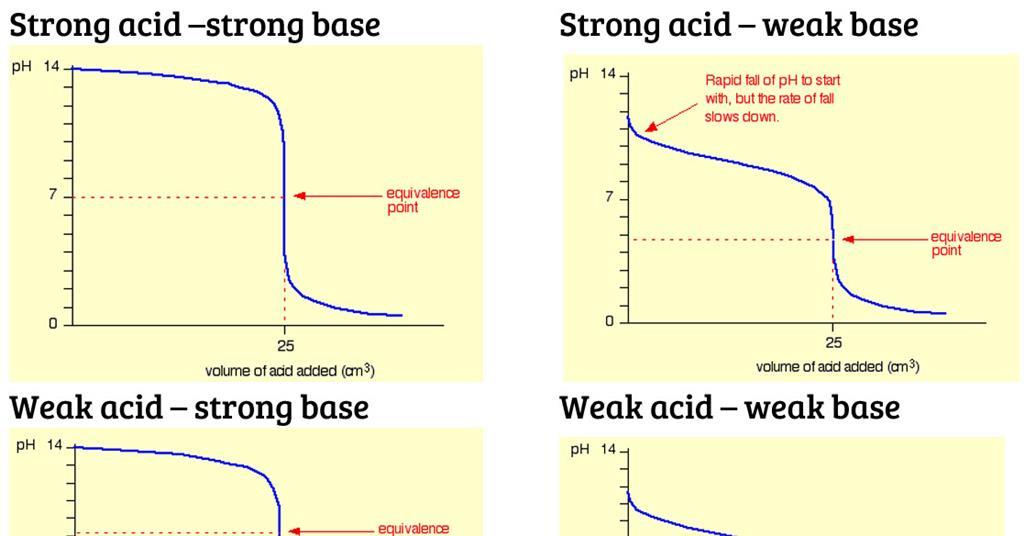
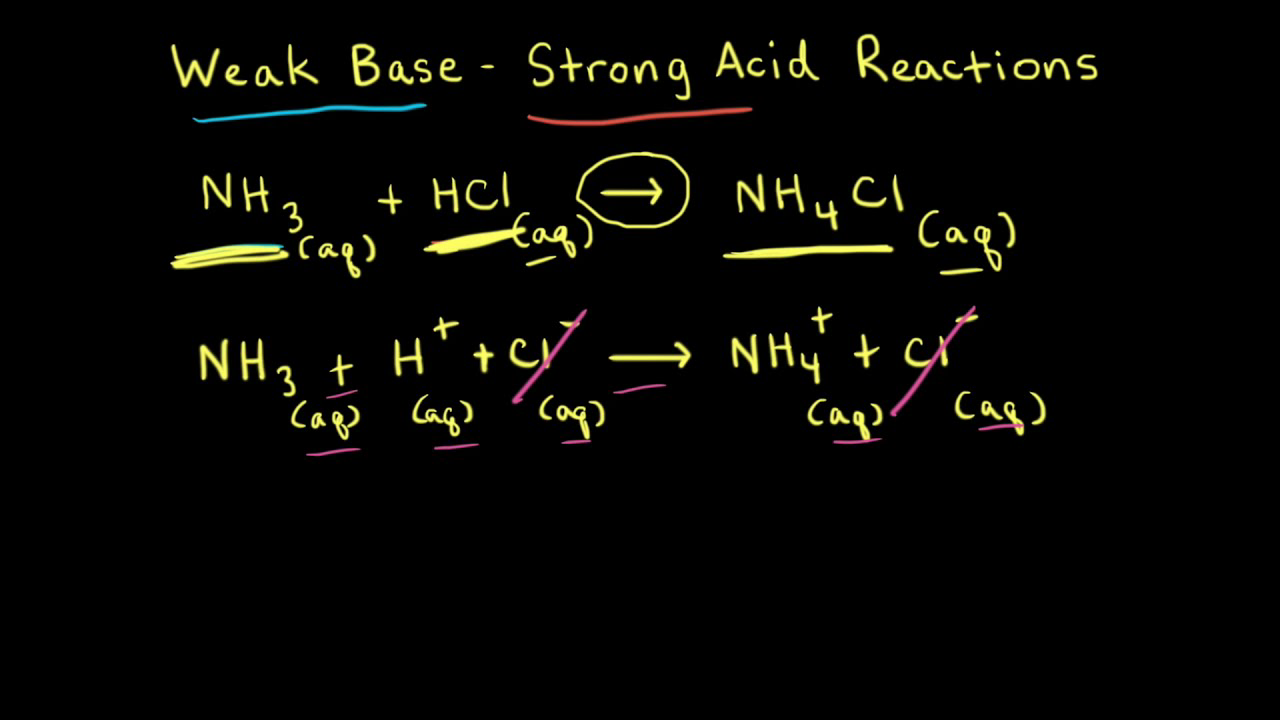
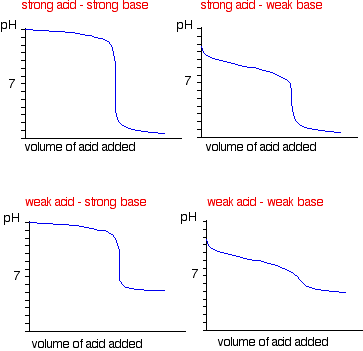
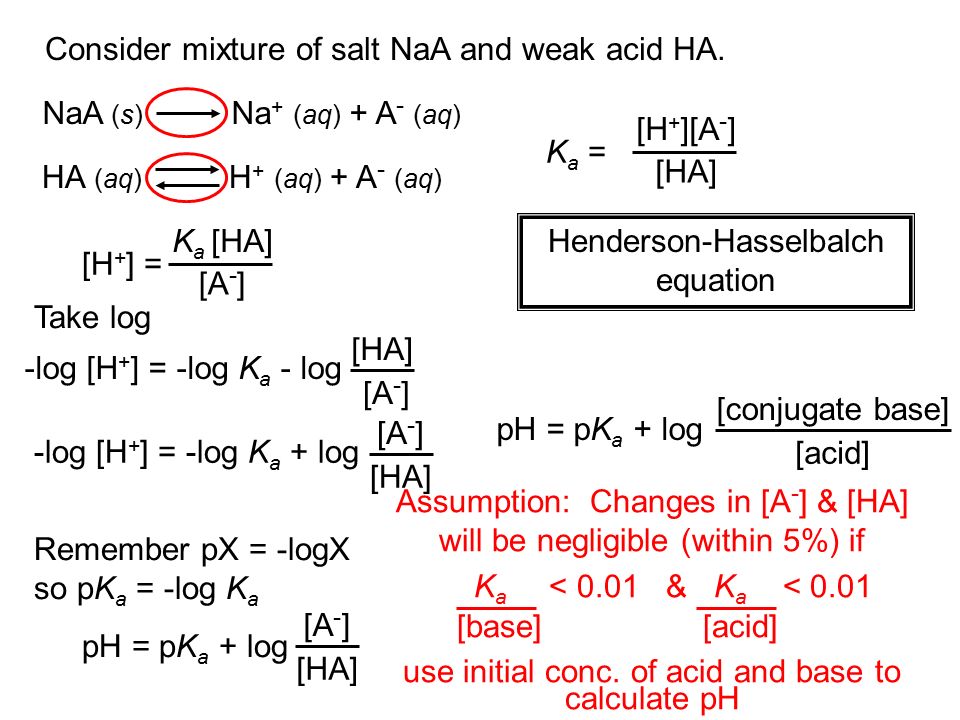
pH of a solution of salt of weak acid and weak base is : pH=1/2pKw+1/2pKa-1/2pKb and that of weak acid and strong base is pH=1/2pKw+1/2pKa+1/2logc For a salt of weak acid and

Calculation of Hydrolysis Constant, Degree of Hydrolysis and pH of Salt Solution - Chemistry, Class 11, Ionic Equilibrium
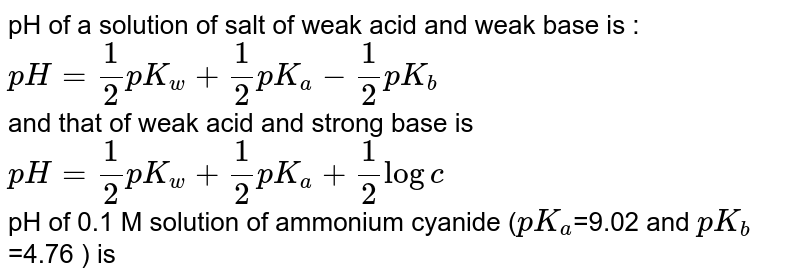
pH of a solution of salt of weak acid and weak base is : pH=1/2pKw+1/2pKa-1/2pKb and that of weak acid and strong base is pH=1/2pKw+1/2pKa+1/2logc For a salt of weak acid and

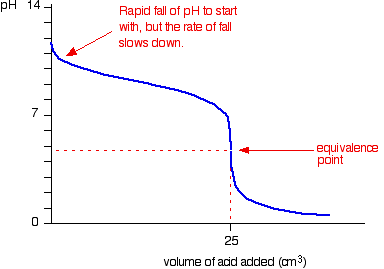
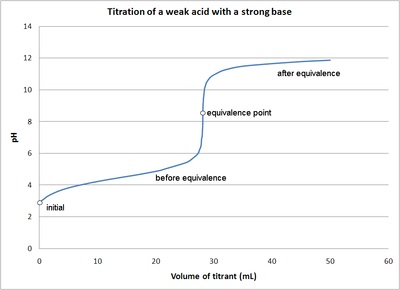
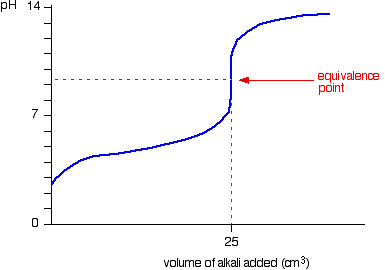
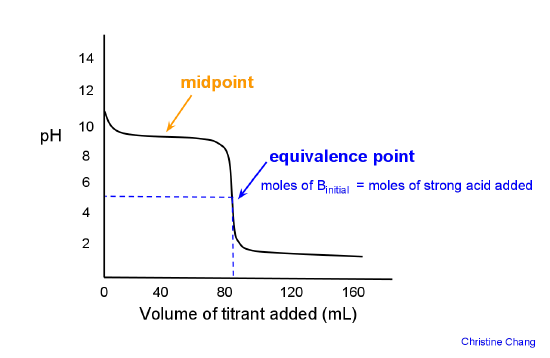
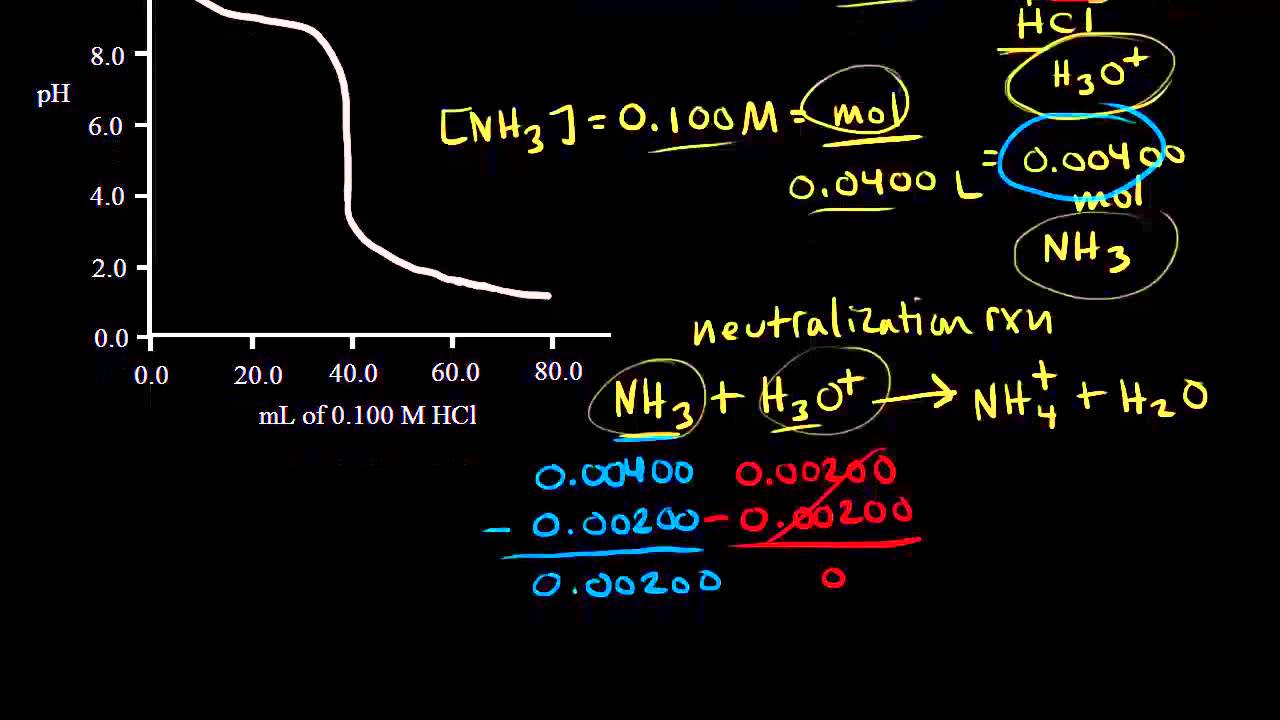
Here is an example of a titration curve, produced when a strong base is added to a strong acid. This curve shows how pH varies as 0.100 M NaOH is added to 50.0 mL of 0.100 M HCl.